Abstract
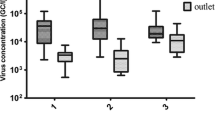
Human bocavirus (HBoV) infections are related to respiratory and gastroenteric diseases. The aim of this study was to investigate the presence of HBoV in both sewage and surface waters in Uruguay. Sixty-eight sewage samples from the cities of Salto, Paysandú, Bella Unión, Fray Bentos, Treinta y Tres and Melo and 36 surface water samples from the cities of Salto, Florida and Santa Lucía were studied. HBoV was screened by multiplex qPCR for the detection of the four subtypes, followed by monoplex qPCRs for the independent quantification of each subtype. A qualitative PCR followed by DNA sequencing and phylogenetic analysis was carried out for molecular characterization of HBoV strains. HBoV was present in a high frequency (69%) in sewage and only one positive sample (3%) was found in surface water. Concerning sewage samples, HBoV1 was detected in 11 (23%) out of the 47 positives samples, with a mean concentration of 8.2 × 104 genomic copies/Liter (gc/L), HBoV3 was detected in 35 (74%) of the positive samples with a mean concentration of 4.1 × 106 gc/L and subtypes 2 and/or 4 were detected in 39 (83%) of the positive samples with a mean concentration of 7.8 × 106 gc/L. After the phylogenetic analysis performed by a Bayesian approach, the four HBoV subtypes were confirmed. This is the first study determining a high frequency of HBoV and the presence of the four HBoV subtypes in aquatic matrices in Latin America, mainly in sewage. Although HBoV was scarcely detected in surface water, a waterborne transmission is likely to occur if people enter in contact with polluted surface waters for recreational activities such as fishing or swimming since an elevated frequency of HBoV was detected in raw sewage which is usually directly discharged into surface waters.


Similar content being viewed by others
References
Albuquerque, M. C. M., Rocha, L. N., Benati, F. J., Soares, C. C., Maranhão, A. G., Ramirez, M. L., et al. (2007). Human bocavirus infection in children with gastroenteritis, Brazil. Emerging Infectious Diseases, 13, 1756–1758.
Allander, T., Tammi, M. T., Eriksson, M., Bjerkner, A., TiveljungLindell, A., & Andersson, B. (2005). Cloning of a human parvovirus by molecular screening of respiratory tract samples. Proceedings of the National academy of Sciences of the United States of America, 102, 12891–12896.
Arthur, J. L., Higgins, G. D., Davidson, G. P., Givney, R. C., & Ratcliff, R. M. (2009). A novel bocavirus associated with acute gastroenteritis in Australian children. PLoS Pathogens, 5, e1000391.
Bibby, K., & Peccia, J. (2013). Identification of viral pathogen diversity in sewage sludge by metagenome analysis. Environmental Science and Technology, 47, 1945–1951.
Blinkova, O., Rosario, K., Li, L., Kapoor, A., Slikas, B., Bernardin, F., et al. (2009). Frequent detection of highly diverse variants of cardiovirus, cosavirus, bocavirus, and circovirus in sewage samples collected in the United States. Journal of Clinical Microbiology, 47, 3507–3513.
Burutarán, L., Lizasoain, A., García, M., Tort, L. F., Colina, R., & Victoria, M. (2015). Detection and molecular characterization of aichivirus 1 in wastewater samples from Uruguay. Food and Environmental Virology, 8, 13–17.
Campe, H., Hartberger, C., & Sing, A. (2008). Role of human bocavirus infections in outbreaks of gastroenteritis. Journal of Clinical Virology, 43, 340–342.
Chhabra, P., Payne, D. C., Szilagyi, P. G., Edwards, K. M., Staat, M. A., Shirley, S. H., et al. (2013). Etiology of viral gastroenteritis in children < 5 years of age in the United States, 2008–2009. Journal of Infectious Diseases, 208, 790–800.
Choi, E. H., Lee, H. J., Kim, S. J., Eun, B. W., Kim, N. H., Lee, J. A., et al. (2006). The association of newly identified respiratory viruses with lower respiratory tract infections in Korean children, 2000–2005. Clinical Infectious Diseases, 43(5), 585–592.
Darriba, D., Taboada, G. L., Doallo, R., & Posada, D. (2012). jModelTest 2: more models, new heuristics and parallel computing. Nature Methods, 9(8), 772.
Edgard, R. C. (2004). MUSCLE: multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Research, 32(5), 1792–1795.
Guido, M., Tumolo, M. R., Verri, T., Romano, A., Serio, F., De Giorgi, M., et al. (2016). Human bocavirus: current knowledge and future challenges. World Journal of Gastroenterology, 22, 8684–8697. https://doi.org/10.3748/wjg.v22.i39.8684.
Guindon, S., & Gascuel, O. (2003). A simple, fast and accurate method to estimate large phylogenies by maximum-likelihood. Systematic Biology, 52, 696–704.
Hamza, I. A., Jurzik, L., Wilhelm, M., & Uberla, K. (2009). Detection and quantification of human bocavirus in river water. Journal of General Virology, 90, 2634–2637.
Hamza, H., Leifels, M., Wilhelm, M., & Hamza, I. A. (2017). Relative abundance of human bocaviruses in urban sewage in Greater Cairo, Egypt. Food and Environmental Virology. https://doi.org/10.1007/s12560-017-9287-3.
Haramoto, E., Katayama, H., Utagawa, E., & Ohgaki, S. (2009). Recovery of human norovirus from water by virus concentration methods. Journal of Virological Methods, 160(1–2), 206–209.
Huelsenbeck, J. P., & Ronquist, F. (2001). MR BAYES: bayesian inference of phylogenetic trees. Bioinformatics, 17, 754–755.
Iaconelli, M., Divizia, M., Della Libera, S., Di Bonito, P., & La Rosa, G. (2016). Frequent detection and genetic diversity of human bocavirus in urban sewage samples. Food and Environmental Virology, 8, 289–295.
Kantola, K., Sadeghi, M., & Antikainen, J. (2010). Real-time quantitative PCR detection of four human bocaviruses. Journal of Clinical Microbiology, 48, 4044–4050.
Kapoor, A., Simmonds, P., Slikas, E., Li, L., Bodhidatta, L., Sethabutr, O., et al. (2010). Human bocaviruses are highly diverse, dispersed, recombination prone, and prevalent in enteric infections. Journal of Infectious Diseases, 201, 1633–1643.
Kapoor, A., Slikas, E., Simmonds, P., Chieochansin, T., Naeem, A., Shaukat, S., et al. (2009). A newly identified bocavirus species in human stool. Journal of Infectious Diseases, 199, 196–200.
Katayama, H., Shimasaki, A., & Ohgaki, S. (2002). Development of a virus concentration method and its application to detection of enterovirus and norwalk virus from coastal seawater. Applied and Environmental Microbiology, 68, 1033–1039.
La Rosa, G., Della Libera, S., Iaconelli, M., Donia, D., Cenko, F., Xhelilaj, G., et al. (2015). Human bocavirus in children with acute gastroenteritis in Albania. Journal of Medical Virology, 88, 906–910.
La Rosa, G., Sanseverino, I., Della Libera, S., Iaconelli, M., Ferrero, V. E. V., Barra Caracciolo, A., et al. (2017). The impact of anthropogenic pressure on the virological quality of water from the Tiber river, Italy. Lett Appl Microbiol. https://doi.org/10.1111/lam.12774.
Lau, S. K., Yip, C. C., Que, T. L., Lee, R. A., Au-Yeung, R. K., Zhou, B., et al. (2007). Clinical and molecular epidemiology of Human Bocavirus in respiratory and fecal samples from children in Hong Kong. Journal of Infectious Diseases, 196, 986–993.
Lizasoain, A., Tort, L. F., García, M., Gómez, M. M., Cristina, J., Leite, J. P., et al. (2015). Environmental assessment of classical human astrovirus in Uruguay. Food and Environmental Virology, 7, 142.
Myrmel, M., Lange, H., & Rimstad, E. (2015). A 1-year quantitative survey of noro-, adeno-, human boca-, and hepatitis e viruses in raw and secondarily treated sewage from two plants in Norway. Food and Environmental Virology, 7, 213–223.
Nawaz, S., Allen, D. J., Aladin, F., Gallimore, C., & Iturriza-Gomara, M. (2012). Human bocaviruses are not significantly associated with gastroenteritis: results of retesting archive DNA from a case control study in the UK. PLoS ONE, 7(7), e41346.
Proenca-Modena, J. L., Gagliardi, T. B., Escremim de Paula, F., Iwamoto, M. A., Criado, M. F., Camara, A. A., et al. (2011). Detection of human bocavirus mRNA in respiratory secretions correlates with high viral load and concurrent diarrhea. PLoS ONE, 6(6), e21083.
Proenca-Modena, J. L., Martinez, M., Amarilla, A. A., Espínola, E. E., Galeano, M. E., Fariña, N., et al. (2013). Viral load of Human Bocavirus-1 in stools from children with viral diarrhoea in Paraguay. Epidemiology and Infection, 141(12), 2576–2580.
Ronquist, F., & Huelsenbeck, J. P. (2003). MrBayes 3: bayesian phylogenetic inference under mixed models. Bioinformatics, 19(12), 1572–1574.
R Core Team (2013). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, URL. http://www.R-project.org/.
Tort, L. F., Victoria, M., Lizasoain, A., García, M., Berois, M., Cristina, J., et al. (2015). Detection of common, emerging and uncommon VP4, and VP7 human group A rotavirus genotypes from urban sewage samples in Uruguay. Food and Environmental Virology, 7, 342–353.
Verbyla, M. E., & Mihelcic, J. R. (2015). A review of virus removal in wastewater treatment pond systems. Water Research, 71, 107–124.
Victoria, M., Tort, L. F., García, M., Lizasoain, A., Maya, L., Leite, J. P., et al. (2014). Assessment of gastroenteric viruses from wastewater directly discharged into Uruguay river, Uruguay. Food and Environmental Virology, 6, 116–124.
Victoria, M., Tort, L. F., Lizasoain, A., García, M., Castells, M., Berois, M., et al. (2016). Norovirus molecular detection in Uruguayan sewage samples reveals a high genetic diversity and GII.4 variant replacement along time. Journal of Applied Microbiology, 120, 1427–1435.
Acknowledgments
We would like to thank the financial support given by the program ‘‘Polo de Desarrollo Universitario (PDU), Universidad de la República (UdelaR), Uruguay’’; project CSIC I+D 2010 and project CSIC I+D 2014, UdelaR. We also thank OSE and the staff of the Virology Section, School of Sciences, UdelaR, Uruguay for their technical assistance. MS is a master student at “Programa de Desarrollo de las Ciencias Básicas - PEDECIBA” and has a scholarship from “Agencia Nacional de Investigación e Innovación - ANII”, Uruguay.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Salvo, M., Lizasoain, A., Castells, M. et al. Human Bocavirus: Detection, Quantification and Molecular Characterization in Sewage and Surface Waters in Uruguay. Food Environ Virol 10, 193–200 (2018). https://doi.org/10.1007/s12560-017-9334-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12560-017-9334-0