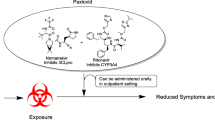
Opinion statement
Purpose of Review With the advent of direct acting antiviral (DAAs) drugs, hepatitis c is now a curabledisease. The human immunodeficiency virus (HIV) and hepatitis c virus (HCV) have similar risk factorsfor acquisition, and thus the co-infected patient population provides its own set of unique challenges. Theclinical course, monitoring and treatment of co-infected patients will be reviewed. Important druginteractions between DAAs and antiretroviral therapies (ART) will be highlighted.
Recent Findings HIV accelerates the natural history of HCV, and liver complications (including end-stageliver disease) resulting from HCV are the most common cause of death in co-infected persons. Five DAAregimens are been well studied in HIV/HCV co-infected patients with sustained virologic responsesbetween 91-100%. Two new DAA regimens have been approved by the FDA and can be used with ART.Some DAA regimens are limited by genotype, underlying renal disease, and decompensated cirrhosis.
Summary This review discusses the impact of HCV/HIV co-infection on morbidity and mortality inHIV-infected persons, focuses on the most current treatment options for HCV, reviews key druginteractions between ART and DAAs, and highlights future treatment options. We recommend thatall co-infected patients be treated for chronic HCV except those whose life expectancy is less than afew years and will not be remediated by HCV treatment.
Similar content being viewed by others
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Global AIDS Update 2016. UNAIDS; 2016.
Mohd Hanafiah K, Groeger J, Flaxman AD, Wiersma ST. Global epidemiology of hepatitis C virus infection: new estimates of age-specific antibody to HCV seroprevalence. Hepatology. 2013;57(4):1333–42.
Sherman KE, Rouster SD, Chung RT, Rajicic N. Hepatitis C virus prevalence among patients infected with human immunodeficiency virus: a cross-sectional analysis of the US adult AIDS Clinical Trials Group. Clin Infect Dis. 2002;34(6):831–7.
Di Martino V, Rufat P, Boyer N, Renard P, Degos F, Martinot-Peignoux M, et al. The influence of human immunodeficiency virus coinfection on chronic hepatitis C in injection drug users: a long-term retrospective cohort study. Hepatology. 2001;34(6):1193–9.
Viral Hepatitis Surveillance Report 2015.
Ly KN, Hughes EM, Jiles RB, Holmberg SD. Rising mortality associated with hepatitis C virus in the United States, 2003-2013. Clin Infect Dis. 2016;62(10):1287.
Kim H, Nance R, Van Rompaey S, Delaney J, Crane H, Cachay E, et al. Poorly controlled HIV infection: an independent risk factor for liver fibrosis. JAIDS J Acquir Immune Defic Syndr. 2016;72(4):437–43.
Rockstroh JK, Mocroft A, Soriano V, Tural C, Losso MH, Horban A, et al. Influence of hepatitis C virus infection on HIV-1 disease progression and response to highly active antiretroviral therapy. J Infect Dis. 2005;192(6):992–1002.
Qurishi N, Kreuzberg C, Lüchters G, Effenberger W, Kupfer B, Sauerbruch T, et al. Effect of antiretroviral therapy on liver-related mortality in patients with HIV and hepatitis C virus coinfection. Lancet. 2003;362(9397):1708–13.
•• Naggie S, Cooper C, Saag M, Workowski K, Ruane P, Towner WJ, et al. Ledipasvir and sofosbuvir for HCV in patients coinfected with HIV-1. N Engl J Med. 2015;373(8):705. This is the Ion 4 study that evaluated use of ledipasvir and sofosbuvir in HIV/HCV co infected patients
• Wyles D, Saag M, Viani RM, Lalezari J, Adeyemi O, Bhatti L, et al. TURQUOISE-I part 1b: ombitasvir/paritaprevir/ritonavir and dasabuvir with ribavirin for hepatitis C virus infection in HIV-1 coinfected patients on darunavir. J Infect Dis. 2017;215(4):599–605. This is the TURQUOISE-1 study that evaluated the use of ombitasvir, paritaprevir, ritonavir and dasabuvir with ribavirin for hepatitis C virus infection in HIV-1 co-infected patients.
•• Wyles D, Bru N, Kottilil S, Daar ES, Ruane P, Workowski K, et al. Sofosbuvir and velpatasvir for the treatment of hepatitis C virus in patients coinfected with human immunodeficiency virus type 1: an open-label, phase 3 study. Clinical Infectious Diseases. 2017:cix260. This is the ASTRAL 5 trial that evaluated sofosbuvir and velpatasvir in co infected patients.
•• Rockstroh JK, Nelson M, Katlama C, Lalezari J, Mallolas J, Bloch M, et al. Efficacy and safety of grazoprevir (MK-5172) and elbasvir (MK-8742) in patients with hepatitis C virus and HIV co-infection (C-EDGE CO-INFECTION): a non-randomised, open-label trial. Lancet HIV. 2015;2(8):e327. This is the C-Edge trial that evaluated efficacy and safety of grazoprevir and elbasvir in co infected patients
•• Osinusi A, Townsend K, Kohli A, Nelson A, Seamon C, Meissner EG, et al. Virologic response following combined ledipasvir and sofosbuvir administration in patients with HCV genotype 1 and HIV co-infection. JAMA. 2015;313(12):1232–9. This is the ERADICATE trial that evaluated virologic response with use of ledipasvir/sofosbuvir in co infected patients
•• Sulkowski M, Hezode C, Gerstoft J, Vierling JM, Mallolas J, Pol S, et al. Efficacy and safety of 8 weeks versus 12 weeks of treatment with grazoprevir (MK-5172) and elbasvir (MK-8742) with or without ribavirin in patients with hepatitis C virus genotype 1 mono-infection and HIV/hepatitis C virus co-infection (C-WORTHY): a randomised, open-label phase 2 trial. Lancet. 2015;385(9973):1087–97. This is the C-WORTHY trial that evaluated use of grazoprevir and elbasvir with or without ribavirin in co infected patients
•• Sulkowski MS, Eron JJ, Wyles D, Trinh R, Lalezari J, Wang C, et al. Ombitasvir, paritaprevir co-dosed with ritonavir, dasabuvir, and ribavirin for hepatitis C in patients co-infected with HIV-1: a randomized trial. JAMA. 2015;313(12):1223–31. This is the TURQUOISE-I 1a trial that evaluated ombitasvir/paritaprevir/ritonavir/dasabuvir and ribavirin in co infected patients
•• Wyles DL, Ruane PJ, Sulkowski MS, Dieterich D, Luetkemeyer A, Morgan TR, et al. Daclatasvir plus sofosbuvir for HCV in patients coinfected with HIV-1. N Engl J Med. 2015;373(8):714. This is the ALLY-2 trial that evaluated use of daclatasvir/sofosbuvir in co infected patients
Guaraldi G, Orlando G, Zona S, Menozzi M, Carli F, Garlassi E, et al. Premature age-related comorbidities among HIV-infected persons compared with the general population. Clin Infect Dis. 2011;53(11):1120–6.
Jacobson IM, Lim JK, Fried MW. American Gastroenterological Association Institute clinical practice update—expert review: care of patients who have achieved a sustained virologic response after antiviral therapy for chronic hepatitis C infection. Gastroenterology. 2017;152(6):1578–87.
Hum J, Jou JH, Green PK, Berry K, Lundblad J, Hettinger BD, et al. Improvement in glycemic control of type 2 diabetes after successful treatment of hepatitis C virus. Diabetes Care. 2017:dc170485.
Cepeda JA, Thomas DL, Astemborski J, Sulkowski MS, Kirk GD, Mehta SH. Increased mortality among persons with chronic hepatitis C with moderate or severe liver disease: a cohort study. Clinical Infectious Diseases. 2017:cix207.
Ajao A, Cao K, Jason M, Bersoff-Matcha SJ, Jones SC, Brinker A, et al. Hepatitis B virus reactivation associated with direct-acting antiviral therapy for chronic hepatitis C virus: a review of cases reported to the U.S. food and drug administration adverse event reporting system.(Original Research)(United States of America)(Author abstract). Ann Intern Med. 2017;166(11):792.
The Strategies for Management of Antiretroviral Therapy (SMART) Study Group. CD4+ count—guided interruption of antiretroviral treatment. N Engl J Med. 2006;355(22):2283–96.
Khatri A, Dutta S, Wang H, Podsadecki T, Trinh R, Awni W, et al. Evaluation of drug-drug interactions between hepatitis C antiviral agents ombitasvir, paritaprevir/ritonavir, and dasabuvir and HIV-1 protease inhibitors. Clin Infect Dis. 2016;62(8):972.
•• Recommendations for testing, managing, and treating hepatitis C [Internet].; 2017 [updated April 27,; cited 07/30/17]. Available from: http://www.hcvguidelines.org. IDSA/AASLD HCV guidelines.
Basu P, Shah NJ, Brown RS. 369 Simeprevir and sofosbuvir with modified doses of ribavirin (RBV) therapy on telaprevir experienced co infected (with HIV) cirrhotics with chronic hepatitis C (CHC). An open label clinical pilot study: STOP C. Gastroenterology. 2015;148(4):979.
Daklinza. Bristol-Myers Squibb. 2017 February.
Zepatier. Merck. 2017.
Harvoni. Gilead. 2017 April.
Epclusa. Gilead. 2017 March.
Viekira. AbbVie. 2017 March.
Olysio. Jenssen. 2017 May.
• Jacobson IM, Lawitz E, Gane EJ, Willems BE, Ruane PJ, Nahass RG, et al. Efficacy of 8 weeks of sofosbuvir, velpatasvir, and voxilaprevir in patients with chronic HCV infection: 2 phase 3 randomized trials. Gastroenterology. 2017. This is the study that showed efficacy of 8 weeks of one of the newest FDA approved regimens sofosbuvir, velpatasvir, and voxilaprevir in patients with chronic HCV.
• Bourlière M, Gordon SC, Flamm SL, Cooper CL, Ramji A, Tong M, et al. Sofosbuvir, velpatasvir, and voxilaprevir for previously treated HCV infection. N Engl J Med. 2017;376(22):2134. This is the study that showed efficacy of sofosbuvir, velpatasvir, and voxilaprevir in DAA treatment experienced HCV patients.
Zeuzem S, Feld JJ, Wang S, Bourliere M, Wedemeyer H, Gane EJ, et al. ENDURANCE-1: efficacy and safety of 8-versus 12-week treatment with ABT-493/ABT-530 in patients with chronic HCV genotype 1 infection. Hepatology; WILEY-BLACKWELL 111 RIVER ST, HOBOKEN 07030-5774, NJ USA; 2016.
Foster G, Gane E, Asatryan A, Pol S, Poordad F, Vierling J, et al. Endurance-3: a phase 3, randomized, open-label, active-controlled study to compare efficacy and safety of ABT-493/ABT-530 to sofosbuvir co-administered with daclatasvir in adults with HCV genotype 3 infection. J Hepatol. 2016;64(2):S292.
Forns X, Lee S, Valdes J, Lens S, Ghalib R, Aguilar H, et al. GS-006-EXPEDITION-I: efficacy and safety of glecaprevir/pibrentasvir in adults with chronic hepatitis C virus genotype 1, 2, 4, 5 or 6 infection and compensated cirrhosis. J Hepatol. 2017;66(1):S4.
Rockstroh J, Lacombe K, Viani RM, Orkin C, Wyles D, Luetkemeyer A, et al. LBP-522-efficacy and safety of glecaprevir/pibrentasvir in patients co-infected with hepatitis C virus and human immunodeficiency virus-1: the EXPEDITION-2 Study. J Hepatol. 2017;66(1):S103.
Lawtiz E, Yoshida EM, Buti M, et al. Safety and efficacy of the fixed-dose combination regimen of MK-3682/grazoprevir/MK-8408 with or without ribavirin in non-cirrhotic or cirrhotic patients with chronic HCV GT1,2 or 3 infection. Gastroenterol Hepatol. 2016;12(Supplement 6):15–6.
Londoo M, Manzardo C, Rimola A, Ruiz P, Costa J, Forner A, et al. IFN-free therapy for HIV/HCV-coinfected patients within the liver transplant setting. J Antimicrob Chemother. 2016;71(11):3195–201.
Grant JL, Hawkins C, Brooks H, Palella FJ Jr, Koppe SW, Abecassis MM, et al. Successful sofosbuvir-based therapy in HIV/HCV co-infected liver transplant recipients with recurrent HCV infection. AIDS. 2016;30(1):93.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Dr. Rebecca Kumar declares that she has no conflict of interest.
Dr. Gayle Balba reports serving as an investigator for Gilead and owning stock in Gilead and GlaxoSmithKline.
Human and animal rights and informed consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
This article is part of the Topical Collection on Hepatitis C
Rights and permissions
About this article
Cite this article
Kumar, R.N., Balba, G.P. Managing the HIV/HCV-Co-Infected Patient in the Direct-Acting Antiviral Era: a Review of Pertinent Drug Interactions. Curr Treat Options Infect Dis 9, 411–424 (2017). https://doi.org/10.1007/s40506-017-0138-4
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40506-017-0138-4