Abstract
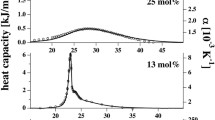
Results obtained via small-angle neutron scattering studies of the influence of calcium ions on the structure and phase transitions of phospholipid membranes are presented. The main phase transition temperature of 1,2-dimyristoyl-sn-glycero-3-phosphatidylcholine (1 wt %) multilamellar vesicles is demonstrated to increase by more than 1°C even when the calcium-ion content of the solution is low (0.1 mM). Detailed analysis of the multilamellar vesicles transition between “bound” and “unbound” state indicates the continuous character of the investigated process in both liquid and gel phases. The critical Ca2+ ion concentrations which initiate the destruction of the multilamellar structures and the formation of unilamellar vesicles are found to be ~0.3 mM in the gel and ~0.4–0.5 mM in the liquid-crystal phases during heating and ~0.5 mM in the phases under study upon cooling.
Similar content being viewed by others
References
R. B. Gennis, Biomembranes: Molecular Structure and Function (Springer, New York, 1989). doi 10.1007/978-1-4757-2065-5
H. C. Lee, R. Aarhus, and T. F. Walseth, Science (Washington, DC) 261 (5119), 352 (1993). doi 10.1126/science.8392749
M. Berridge, P. Lipp, and M. Bootman, Curr. Biol. 9, 157 (1999). doi 10.1016/S0960-9822(99)80101-8
O. H. Petersen, M. Michalak, and A. Verkhratsky, Cell Calcium 38, 161 (2005). doi 10.1016/j.ceca.2005.06.023
A. G. Lee, Biochim. Biophys. Acta 1666, 62 (2004). doi 10.1016/j.bbamem.2004.05.012
M. Rappolt, G. Pabst, H. Amenitsch, and P. Laggner, Colloids Surf., A 183–185, 171 (2001). doi 10.1016/S0927-7757(01)00568-4
R. A. Böckmann, A. Hac, T. Heimburg, and H. Grubmüller, Biophys. J. 85, 1647 (2003). doi 10.1016/s0006-3495(03)74594-9
H. I. Petrache, I. Kimchi, D. Harries, and V. A. Parsegian, J. Am. Chem. Soc. 127, 11546 (2005). doi 10.1021/ja052549+
H. I. Petrache, S. Tristram-Nagle, D. Harries, N. Kucerka, J. F. Nagle, and V. A. Parsegian, J. Lipid Res. 47, 302 (2006). doi 10.1194/jlr.M500401-JLR200
D. P. Kharakoz, Biosci. Rep. 21, 801 (2006). doi 10.1023/A:1015588825142
J. Seelig, P. M. Macdonald, and P. G. Scherer, Biochemistry 26 (24), 7535 (1987). doi 10.1021/bi00398a001
J. N. Sachs, H. Nanda, H. I. Petrache, and T. B. Woolf, Biophys. J. 86, 3772 (2004). doi 10.1529/biophysj. 103.035816
S. A. Pandit, D. Bostick, and M. L. Berkowitz, Biophys. J. 84, 3743 (2003). doi 10.1016/S0006-3495(03)75102-9
R. A. Böckmann and H. Grubmüller, Angew. Chem., Int. Ed. Engl. 43, 1021 (2004). doi 10.1002/anie.200352784
D. Uhríková, J. Teixeira, A. Lengyela, L. Almásy, and P. Balgavý, J. Spectrosc. 21, 43 (2007). doi 10.1155/2007/576282
G. Pabst, N. Kucerka, M.-P. Nieh, M. C. Rheinstadter, and J. Katsaras, Chem. Phys. Lipids 163, 460 (2010). doi 10.1016/j.chemphyslip.2010.03.010
D. P. Kharakoz, Biosci. Rep. 21 (6), 801 (2001). doi 10.1023/A:1015588825142
Yu. E. Gorshkova and O. I. Ivankov, J. Optoelectron. Adv. Mater. (2016) (in press).
D. Chapman, W. Peel, B. Kingston, and T. Lilley, Biochim. Biophys. Acta 464, 260 (1977). doi 10.1016/0005-2736(77)90002-5
R. Koynova and M. Caffrey, Biochim. Biophys. Acta 1376, 91 (1998). doi 10.1016/S0304-4157(98)00006-9
S. G. Black and G. S. Dixon, Biochemistry 20 (23), 6740 (1981). doi 10.1021/bi00526a033
N. L. Yamada, H. Seto, T. Takeda, M. Naga, Y. Kawabata, and K. Inoue, J. Phys. Soc. Jpn. 74, 2853 (2005). doi 10.1143/JPSJ.74.2853
R. Lipowsky and B. Zelinska, Phys. Rev. Lett. 62, 1572 (1989). doi 10.1103/PhysRevLett.62.1572
W. Helfrich, J. Phys. II 3, 385 (1993). doi 10.1051/jp2:1993100
J. N. Izraelachvili, J. Phys. Chem. 96, 520 (1992). doi 10.1021/j100181a007
A. I. Kuklin, A. Kh. Islamov, and V. I. Gordeliy, Neutron News 16 (3), 16 (2005). doi 10.1080/10448630500454361
A. G. Soloviev, T. N. Murugova, A. Kh. Islamov, and A. I. Kuklin, J. Phys.: Conf. Ser. 351, 012027 (2012). doi 10.1088/1742-6596/351/1/012027
Yu. M. Ostanevich, Makromol. Chem., Macromol. Symp. 15, 91 (1988). doi 10.1002/masy.19880150107
V. I. Gordeliy, V. Cherezov, and J. Teixeira, Phys. Rev. E: Stat., Nonlinear, Soft Matter Phys. 72, 1 (2005). doi 10.1103/PhysRevE.72.061913
Yu. E. Gorshkova, J. Optoelectron. Adv. Mater. 17 (9–10), 1532 (2015).
L. A. Feigin and D. I. Svergun, Structure Analysis by Small-angle X-ray and Neutron Scattering (Plenum, New York, 1987). doi 10.1002/actp.1989.010400317
V. I. Gordeliy, L. V. Golubchikova, A. I. Kuklin, A. G. Syrykh, and A. Watts, Prog. Colloid Polym. Sci. 93, 252 (1993). doi 10.1007/BFb0118537
D. Uhríková, N. Kucerka, J. Teixeira, V. Gordeliy, and P. Balgavý, Chem. Phys. Lipids 155, 80 (2008). doi 10.1016/j.chemphyslip.2008.07.010
Y. Inoko, T. Yamaguchi, K. Furuya, and T. Mitsui, Biochim. Biophys. Acta 413, 24 (1975). doi 10.1016/0005-2736(75)90055-3
L. J. Lis, V. A. Parsegian, and R. P. Rand, Biochemistry 20, 1761 (1981). doi 10.1021/bi00510a010
Y. Izumitani, J. Colloid Interface Sci. 166, 143 (1994). doi 10.1006/jcis.1994.1281
Y. Izumitani, J. Colloid Interface Sci. 182, 6 (1996). doi 10.1006/jcis.1996.0431
K. Akashi, H. Miyata, H. Itoh, and K. Kinosita, Biophys. J. 74, 2973 (1998). doi 10.1016/S0006-3495(98)78004-X
J. F. Nagle and S. Tristram-Nagle, Biochim. Biophys. Acta 1469, 159 (2000). doi 10.1016/S0304-4157(00)00016-2
H. Ohshima, Y. Inoko, and T. Mitsui, J. Colloid Interface Sci. 86 (1), 57 (1982). doi 10.1016/0021-9797(82)90041-8
H. Hauser, Phospholipid Handbook (Marcel Dekker, New York, 1993).
Author information
Authors and Affiliations
Corresponding author
Additional information
Original Russian Text © Yu.E. Gorshkova, A.I. Kuklin, V.I. Gordeliy, 2016, published in Poverkhnost’, 2016, No. 11, pp. 34–45.
Rights and permissions
About this article
Cite this article
Gorshkova, Y.E., Kuklin, A.I. & Gordeliy, V.I. Structure and phase transitions of DMPC multilamellar vesicles in the presence of Ca2+ ions. J. Surf. Investig. 11, 27–37 (2017). https://doi.org/10.1134/S1027451016050499
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1027451016050499