Abstract
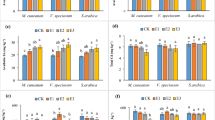
This study was conducted to assess the hyperaccumulation and phytoremediation potential of copper (Cu) and lead (Pb) in Hardy ‘Limelight’ Hydrangea (Hydrangea paniculata) and the common sunflower (Helianthus annuus). The study also investigated the capacity of these two plants to transpire the metals in a temperature-controlled greenhouse. Plants were grown for 4 weeks and periodically watered with known elemental concentrations of copper oxide nanoparticles, copper sulfate, and lead nitrate. Both H. annuus and H. paniculata accumulated significant amounts of Cu and Pb to be classified as hyperaccumulator species. H. annuus took up significant amounts of Cu in the shoots, specifically the leaves (Cu max. = 1368 ppm), and easily translocated it from stem to leaf (translocation factor (TF) ranged from 2.7 to 81.0). Pb was not as easily taken up and translocated (TF = 0.6) as Cu was by this species. H. paniculata took up Cu and Pb in high concentrations but preferentially stored more metals in the stems (Cu max. = 1757 ppm; Pb max. = 780 ppm) than in the leaves (Cu max. = 126 ppm; Pb max. = 35 ppm). The translocation ability of H. paniculata was much lower for both metals compared to H. annuus. Both Cu and Pb transpired from H. annuus at concentrations of 0.04 and 0.005 ppm, respectively.













Similar content being viewed by others
References
Adesodun, J., Atayese, M., Agbaje, T., Osadiaye, B., Mafe, O., & Soretire, A. (2010). Phytoremediation potentials of sunflowers (Tithonia diversifolia and Helianthus annuus) for metals in soils contaminated with zinc and lead nitrates. Water, Air, and Soil Pollution, 207, 195–201.
Baker, A. J. M., & Brooks, R. R. (1989). Terrestrial higher plants which hyperaccumulate metallic elements—a review of their distribution, ecology and phytochemistry. Biorecovery, 1, 81–126.
Baker, A. J. M., Mcgrath, S. P., Reeves, R. D., & Smith, J. A. C. (2000). Metal hyperaccumulator plants: a review of the ecology and physiology of a biological resource for phytoremediation of metal-polluted soils. In N. Terry, G. Banuelos, & J. Vangronsveld (Eds.), Phytoremediation of contaminated soil and water (pp. 85–107). Boca Raton: Lewis.
Chaney, R. L., Malik, M., Li, Y. M., Brown, S. L., Brewer, E. P., Angle, J. S., & Baker, A. L. M. (1997). Phytoremediation of soil metals. Current Opinion in Biotechnology, 8, 279–284.
Duran, N., Marcato, P. D., De Souza, G. I. H., Alves, O. L., & Esposito, E. (2007). Antibacterial effect of silver nanoparticles produced by fungal processes on textile fabrics and their effluent treatment. Journal of Biomedical Nanotechnology, 3(1), 203–208.
Fabrega, J., Luoma, S. N., Tyler, C. R., Galloway, T. S., & Lead, J. R. (2011). Silver nanoparticles: behaviour and effects in the aquatic environment. Environment International, 37(2), 517–531.
Fernandes, J. C., & Henriques, F. S. (1991). Biochemical, physiological, and structural effects of excess copper in plants. The Botanical Review, 57, 246–273.
Garbisu, C., & Alkorta, I. (2003). Basic concepts on heavy metal soil bioremediation. The European Journal of Mineral Processing and Environmental Protection, 3, 58–66.
Hamvumba, R., Mataa, M., & Mweetwa, A. M. (2014). Evaluation of sunflower (Helianthus annuus L.), sorghum (Sorghum bicolor L.), and Chinese cabbage (Brassica chinensis) for phytoremediation of lead contaminated soils. Environment and Pollution, 3(2), 65–73.
Huang, J. W., Chen, J., Berti, W. R., & Cunningham, S. D. (1997). Phytoremediation of lead-contaminated soils: role of synthetic chelates in lead phytoextraction. Environmental Science and Technology, 31, 800–805.
Ikenaka, Y., Nakayama, S. M. M., Muzandu, K., Choongo, K., Teraoka, H., Mizuno, N., & Ishizuka, M. (2010). Heavy metal contamination of soil and sediment in Zambia. African Journal of Environmental Science and Technology, 4(11), 729–739.
Jain, D., Daima, H. K., Kachhwaha, S., & Kothari, S. L. (2009). Synthesis of plant-mediated silver nanoparticles using papaya fruit extract and evaluation of their anti microbial activities. Digest Journal of Nanomaterials and Biostructures, 4(3), 557–563.
Jansen, S., Broadley, M. R., Robbrecht, E., & Smets, E. (2002). Aluminum hyperaccumulation in angiosperms: a review of its phylogenetic significance. The Botanical Review, 68(2), 235–269.
Kastratovic, V., Krivokapic, S., Bigovic, M., Durovic, D., & Blagojevic, N. (2014). Bioaccumulation and translocation of heavy metals by Ceratophyllum demersum from the Skadar Lake, Montenegro. Journal of the Serbian Chemical Society, 79(11), 1445–1460.
Kochian, L. V., Pence, N. S., Lethan, D. L. D., Pineros, M. A., Magalhaes, J. V., Hoekenga, O. A., & Garvin, D. F. (2002). Mechanisms of metal resistance in plants: aluminum and heavy metals. Plant and Soil, 247, 109–119.
Leteinturier, B., Laroche, J., Matera, J., & Malaisse, F. (2001). Reclamation of lead/zinc processing wastes at Kabwe, Zambia: a phytogeochemical approach. South African Journal of Science, 97, 624–627.
Mclean, J. E., & Bledsoe, B. E. (1992). Ground water issue: behavior of metals in soils. EPA/540/S-92/018. Washington, DC: Office of Research and Development.
Missouri Botanical Document. (2016). Hydrangea paniculata. Resource document. Missouri Botanical Garden. http://www.missouribotanicalgarden.org/PlantFinder/PlantFinderDetails.aspx?kempercode=c313. Accessed 22 August 2016.
Mudgal, V., Madaan, N., & Mudgal, A. (2010). Heavy metals in plants: phytoremediation: plants used to remediate heavy metal pollution. Agriculture and Biology Journal of North America, 1, 40–46.
Prasad, M. N. V., & Freitas, H. M. O. (2003). Metal accumulation in plants: biodiversity prospecting for phytoremediation technology. Electronic Journal of Biotechnology, 6(3).
Rahman, M., Azirun, S., & Boyce, A. (2013). Enhanced accumulation of copper and lead in amaranth (Amaranthus paniculatus), Indian mustard (Brassica juncea), and sunflower (Helianthus annuus). Institute of Biological Sciences, 8(5), 1–9.
Schmidt, U. (2003). Enhancing phytoextraction: the effect of chemical soil manipulation on mobility, plant accumulation, and leaching of heavy metals. Journal of Environmental Quality, 32, 1939–1954.
Schrick, B., Hydutsky, B. W., Blough, J. L., & Mallouk, T. E. (2004). Delivery vehicles for zerovalent metal nanoparticles in soil and groundwater. Chemistry of Materials, 16, 2187–2193.
Shah, V., & Belozerova, I. (2008). Influence of metal nanoparticles on the soil microbial community and germination of lettuce seeds. Water, Air, and Soil Pollution, 197, 143–148.
Sracek, O., Kribek, B., Mihaljevic, M., Majer, V., Veselovsky, F., Vencelides, Z., & Nyambe, I. (2012). Mining-related contamination of surface water and sediments of the Kafue River drainage system in the Copperbelt District, Zambia: an example of a high neutralization capacity system. Journal of Geochemical Exploration, 112, 174–188.
Tangahu, B., Abdullah, S., Basri, H., Idris, M., Anuar, N., & Mukhlisin, M. (2011). A review on heavy metals (As, Pb, and Hg) uptake by plants through phytoremediation. International Journal of Chemical Engineering. doi:10.1155/2011/939161.
Wigginton, N. S., Haus, K. L., & Hochella, M. F., Jr. (2007). Aquatic environmental nanoparticles. Journal of Environmental Monitoring, 9(12), 1306–1316.
Wu, F. Y., & Sun, E. J. (1998). Effects of copper, zinc, nickel, chromium, and lead on the growth of water convolvulus in water culture. Journal of Environmental Protection, 21, 63–72.
Yruela, I. (2009). Copper in plants: acquisition, transport and interactions. Functional Plant Biology, 36, 409–430.
Acknowledgements
We thank Alyssa Thomson and Megan Corley for their laboratory and greenhouse assistance. This research was supported by the Department of Biological and Environmental Sciences at Georgia College and State University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Forte, J., Mutiti, S. Phytoremediation Potential of Helianthus annuus and Hydrangea paniculata in Copper and Lead-Contaminated Soil. Water Air Soil Pollut 228, 77 (2017). https://doi.org/10.1007/s11270-017-3249-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11270-017-3249-0