Abstract
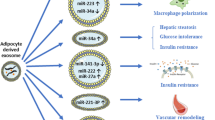
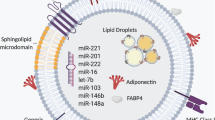
We recently reported that stressed adipocytes release extracellular vesicles (EVs) that act as “find-me” signals to promote macrophage migration and activation. In this study, we performed a comprehensive characterization of stressed adipocyte-derived EVs, assessing their antigenic composition, lipidomics, and RNA profiles. Perilipin A was identified as one of the adipose-specific proteins and studied as a potential novel biomarker to detect adipocyte-derived EVs in circulation. Circulating EVs were significantly increased in mice with diet-induced obesity (DIO) and in obese humans with metabolic syndrome compared to lean controls. This increase was associated with decreased glucose tolerance in the DIO mice and metabolic dysfunction, elevated insulin, and homeostatic model assessment of insulin resistance (HOMA-IR) in the obese humans. EVs from both DIO mice and obese humans were enriched in perilipin A, a central gatekeeper of the adipocyte lipid storehouse and a marker of adipocyte differentiation. In obese humans, circulating levels of EVs enriched in perilipin A were dynamic, decreasing 35 % (p < 0.05) after a 3-month reduced calorie diet intervention. This translational study provides an extensive characterization of adipocyte-derived EVs. The findings identify perilipin A as a novel biomarker of circulating EVs of adipocyte origin and support the development of circulating perilipin A-positive EVs as indicators of adipose tissue health.
Key message
• Extensive characterization of 3T3L1 EVs identified perilipin A in their composition.
• Circulating EVs are elevated in obese mice and associated with glucose intolerance.
• Circulating EVs are elevated in obese human and correlated with metabolic factors.
• Perilipin A and EV levels are increased in the circulation of obese mice and human.
• Circulating EV and perilipin A levels decrease with low calorie intervention.




Similar content being viewed by others
References
Staels B (2006) When the clock stops ticking, metabolic syndrome explodes. Nat Med 12:54–55, discussion 55
Weiss R, Dziura J, Burgert TS, Tamborlane WV, Taksali SE, Yeckel CW, Allen K, Lopes M, Savoye M, Morrison J et al (2004) Obesity and the metabolic syndrome in children and adolescents. N Engl J Med 350:2362–2374
Browning JD, Horton JD (2004) Molecular mediators of hepatic steatosis and liver injury. J Clin lnvest 114:147–152
Tilg H, Moschen AR (2008) Inflammatory mechanisms in the regulation of insulin resistance. Mol Med 14:222–231
Postic C, Girard J (2008) Contribution of de novo fatty acid synthesis to hepatic steatosis and insulin resistance: lessons from genetically engineered mice. J Clin lnvest 118:829–838
Furukawa S, Fujita T, Shimabukuro M, Iwaki M, Yamada Y, Nakajima Y, Nakayama O, Makishima M, Matsuda M, Shimomura I (2004) Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin lnvest 114:1752–1761
Gornicka A, Fettig J, Eguchi A, Berk MP, Thapaliya S, Dixon LJ, Feldstein AE (2012) Adipocyte hypertrophy is associated with lysosomal permeability both in vivo and in vitro: role in adipose tissue inflammation. Am J Physiol Endocrinol Metab 303:E597–606
Neels JG, Olefsky JM (2006) Inflamed fat: what starts the fire? J Clin lnvest 116:33–35
Schenk S, Saberi M, Olefsky JM (2008) Insulin sensitivity: modulation by nutrients and inflammation. J Clin lnvest 118:2992–3002
Wellen KE, Hotamisligil GS (2005) Inflammation, stress, and diabetes. J Clin lnvest 115:1111–1119
Weisberg SP, McCann D, Desai M, Rosenbaum M, Leibel RL, Ferrante AW Jr (2003) Obesity is associated with macrophage accumulation in adipose tissue. J Clin lnvest 112:1796–1808
Weisberg SP, Hunter D, Huber R, Lemieux J, Slaymaker S, Vaddi K, Charo I, Leibel RL, Ferrante AW Jr (2006) CCR2 modulates inflammatory and metabolic effects of high-fat feeding. J Clin lnvest 116:115–124
Hevener AL, Olefsky JM, Reichart D, Nguyen MT, Bandyopadyhay G, Leung HY, Watt MJ, Benner C, Febbraio MA, Nguyen AK et al (2007) Macrophage PPAR gamma is required for normal skeletal muscle and hepatic insulin sensitivity and full antidiabetic effects of thiazolidinediones. J Clin lnvest 117:1658–1669
Lumeng CN, Bodzin JL, Saltiel AR (2007) Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J Clin lnvest 117:175–184
Eguchi A, Mulya A, Lazic M, Radhakrishnan D, Berk MP, Povero D, Gornicka A, Feldstein AE (2015) Microparticles release by adipocytes act as “find-me” signals to promote macrophage migration. PLoS One 10, e0123110. doi:10.1371/journal.pone.0123110
Lee MJ, Wu Y, Fried SK (2012) A modified protocol to maximize differentiation of human preadipocytes and improve metabolic phenotypes. Obesity 20:2334–2340
Lee MJ, Wu Y, Fried SK (2013) Adipose tissue heterogeneity: implication of depot differences in adipose tissue for obesity complications. Mol Aspects Med 34:1–11
Yoshizaki T, Milne JC, Imamura T, Schenk S, Sonoda N, Babendure JL, Lu JC, Smith JJ, Jirousek MR, Olefsky JM (2009) SIRT1 exerts anti-inflammatory effects and improves insulin sensitivity in adipocytes. Mol Cell Biol 29:1363–1374
Guttman M, Betts GN, Barnes H, Ghassemian M, van der Geer P, Komives EA (2009) Interactions of the NPXY microdomains of the low density lipoprotein receptor-related protein 1. Proteomics 9:5016–5028
McCormack AL, Schieltz DM, Goode B, Yang S, Barnes G, Drubin D, Yates JR 3rd (1997) Direct analysis and identification of proteins in mixtures by LC/MS/MS and database searching at the low-femtomole level. Anal Chem 69:767–776
Paoletti AC, Parmely TJ, Tomomori-Sato C, Sato S, Zhu D, Conaway RC, Conaway JW, Florens L, Washburn MP (2006) Quantitative proteomic analysis of distinct mammalian mediator complexes using normalized spectral abundance factors. Proc Natl Acad Sci U S A 103:18928–18933
Quehenberger O, Armando AM, Brown AH, Milne SB, Myers DS, Merrill AH, Bandyopadhyay S, Jones KN, Kelly S, Shaner RL et al (2010) Lipidomics reveals a remarkable diversity of lipids in human plasma. J Lipid Res 51:3299–3305
Quehenberger O, Yamashita T, Armando AM, Dennis EA, Palinski W (2011) Effect of gestational hypercholesterolemia and maternal immunization on offspring plasma eicosanoids. Am J Obstet Gynecol 205(156):e115–125
Mitchell PS, Parkin RK, Kroh EM, Fritz BR, Wyman SK, Pogosova-Agadjanyan EL, Peterson A, Noteboom J, O’Briant KC, Allen A et al (2008) Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci U S A 105:10513–10518
Liu G, Abraham E (2013) MicroRNAs in immune response and macrophage polarization. Arterioscler, Thromb, Vasc Biol 33:170–177
Gray WD, Mitchell AJ, Searles CD (2015) An accurate, precise method for general labeling of extracellular vesicles. MethodsX 2:360–367
Silverman JM, Reiner NE (2011) Exosomes and other microvesicles in infection biology: organelles with unanticipated phenotypes. Cell Microbiol 13:1–9
Sears DD, Miles PD, Chapman J, Ofrecio JM, Almazan F, Thapar D, Miller YI (2009) 12/15-lipoxygenase is required for the early onset of high fat diet-induced adipose tissue inflammation and insulin resistance in mice. PLoS One 4, e7250. doi:10.1371/journal.pone.0007250
Chapman J, Miles PD, Ofrecio JM, Neels JG, Yu JG, Resnik JL, Wilkes J, Talukdar S, Thapar D, Johnson K et al (2010) Osteopontin is required for the early onset of high fat diet-induced insulin resistance in mice. PLoS One 5, e13959. doi:10.1371/journal.pone.0013959
Bickel PE, Tansey JT, Welte MA (2009) PAT proteins, an ancient family of lipid droplet proteins that regulate cellular lipid stores. Biochim Biophys Acta 1791:419–440
Fujimoto T, Parton RG (2011) Not just fat: the structure and function of the lipid droplet. Cold Spring Harbor Perspect Biol 3:a004838
Nowicka P, Santoro N, Liu H, Lartaud D, Shaw MM, Goldberg R, Guandalini C, Savoye M, Rose P, Caprio S (2011) Utility of hemoglobin A(1c) for diagnosing prediabetes and diabetes in obese children and adolescents. Diabetes Care 34:1306–1311
Mathurin P, Bataller R (2015) Trends in the management and burden of alcoholic liver disease. J Hepatol 62:S38–46
Brasaemle DL (2007) Thematic review series: adipocyte biology. The perilipin family of structural lipid droplet proteins: stabilization of lipid droplets and control of lipolysis. J Lipid Res 48:2547–2559
Kern PA, Di Gregorio G, Lu T, Rassouli N, Ranganathan G (2004) Perilipin expression in human adipose tissue is elevated with obesity. J Clin Endocrinol Metab 89:1352–1358
Feng D, Tang Y, Kwon H, Zong H, Hawkins M, Kitsis RN, Pessin JE (2011) High-fat diet-induced adipocyte cell death occurs through a cyclophilin D intrinsic signaling pathway independent of adipose tissue inflammation. Diabetes 60:2134–2143
Kolak M, Westerbacka J, Velagapudi VR, Wagsater D, Yetukuri L, Makkonen J, Rissanen A, Hakkinen AM, Lindell M, Bergholm R et al (2007) Adipose tissue inflammation and increased ceramide content characterize subjects with high liver fat content independent of obesity. Diabetes 56:1960–1968
Bastos-Amador P, Royo F, Gonzalez E, Conde-Vancells J, Palomo-Diez L, Borras FE, Falcon-Perez JM (2012) Proteomic analysis of microvesicles from plasma of healthy donors reveals high individual variability. J Proteomics 75:3574–3584
Ostergaard O, Nielsen CT, Iversen LV, Jacobsen S, Tanassi JT, Heegaard NH (2012) Quantitative proteome profiling of normal human circulating microparticles. J Proteome Res 11:2154–2163
Yracheta JM, Alfonso J, Lanaspa MA, Roncal-Jimenez C, Johnson SB, Sanchez-Lozada LG, Johnson RJ (2015) Hispanic Americans living in the United States and their risk for obesity, diabetes and kidney disease: genetic and environmental considerations. Postgrad Med 127:503–510
Barteneva NS, Fasler-Kan E, Bernimoulin M, Stern JN, Ponomarev ED, Duckett L, Vorobjev IA (2013) Circulating microparticles: square the circle. BMC Cell Biol 14:23
Acknowledgments
The authors would like to thank Dr. Marilyn Farquhar (University of California, San Diego, UCSD) for the use of the electron microscopy facility; Timo Meerloo for electron microscopy sample preparation; Dr. Majid Ghassemian of the Biomolecular and Proteomics Mass Spectrometry facility (UCSD) for the proteomics study; Dr. Gary Hardiman, director of UCSD Biomedical Genomics Facility (BIOGEM); and Dr. Roman Sasik for analysis of the sequencing study. We acknowledge One World Lab for the assistance with selecting antibodies and providing test size quantities. This work was supported by NIH grant (U01 AA022489) and (DK082451) to AEF, Gilead research scholars program in liver disease to AE, and P. Robert Majumder Charitable Foundation to DDS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
DDS was a paid consultant of Zone Labs, Inc. prior to the initiation of the human diet intervention study for which Zone Labs, Inc. donated food supplies.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Table 1
Proteomic analysis of total protein composition in palmitic-acid induced sadEV. (XLSX 22 kb)
Supplementary Table 2
Complete list of fatty acids and their bioactive lipid mediators in sadEVs. Unit: pmol/106 sadEV (XLSX 47 kb)
Supplementary Table 3
Complete list of miRNAs in sadEVs and control sadEVs and their respective parent adipocytes by RNA sequencing. (with a lower threshold of >5 raw counts for positive results). (XLS 82 kb)
Supplementary Table 4
Metabolic parameters at baseline and post-calorie restriction. (DOCX 16 kb)
ESM 5
(DOCX 479 kb)
Rights and permissions
About this article
Cite this article
Eguchi, A., Lazic, M., Armando, A.M. et al. Circulating adipocyte-derived extracellular vesicles are novel markers of metabolic stress. J Mol Med 94, 1241–1253 (2016). https://doi.org/10.1007/s00109-016-1446-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-016-1446-8