Abstract
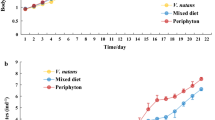
Laboratory cultivation experiments were carried out in order to determine the response of D. galeata to food conditions and dissolved chemicals in the water collected in three contrasting sites of the Rímov Reservoir. There were two types of cultivation media: (1) lake water with natural seston, filtered through a 40 μm mesh size sieve, (2) lake water filtered through a membrane filter 0.45 μm (or 0.22 μm), with Scenedesmus subspicatus culture added to a concentration of 1.0 mg POC l−1. In the treatment with natural seston, the parameters investigated (body length, filtering setae length, clutch size, egg volume and postembryonic development time) were determined mainly by the food level, which was higher in the upper part of the reservoir than near the dam. In one series, significantly larger clutches and smaller eggs together with longer postembryonic development (PED) were detected in daphnids cultivated in hypolimnetic water, compared to those in epilimnetic water. As particulate carbon concentrations were similar, this indicates that in that case food quality played a role shaping life histories in Daphnia. In some of the experiments with the filtered water, there was a detectable effect of dissolved chemical(s), which induced shorter PED and smaller primiparae in the animals cultivated in water from the upper part of the reservoir.
Similar content being viewed by others
References
Bourrelly, P., 1948. L'Algothéque du Laboratoire de Cryptogamie de Muséum. Paris Mus. nat. d'hist.-nat.: 14 pp.
Burns, C.W., 1995. Effects of crowding and different food levels on growth and reproductive investment of Daphnia. Oecologia 101: 234–244.
Carmichael, W. W., 1994. Toxins of cyanobacteria. Sci. am. 270: 78–86.
Dodson, S. I., 1989. The ecological role of chemical stimuli for the zooplankton: predator induced morphology in Daphnia. Oecologia 78: 361–367.
Green, J., 1956. Growth, size and reproduction in Daphnia (Crustacea: Cladocera). Proc. Zool. Soc. London 126: 173–204.
Gulati, R. D. & W. R. DeMott, 1997. The role of food quality for zooplankton: remarks on the state-of-the-art, perspectives and priorities. Freshwat. Biol. 38: 753–768.
Hanazato, T. & S. I. Dodson, 1992. Complex effects of a kairomone of Chaoborus and an insecticide on Daphnia pulex. J. Plankton Res. 14: 1743–1755.
Hejzlar, J. & M. Straškraba, 1989. On the horizontal distribution of limnological variables in Řimov and other stratified Czechoslovak reservoirs. Arch. Hydrobiol. Beih. Ergebn. Limnol. 33: 41–55.
Hessen, D. O. & T. Andersen, 1990. Bacteria as a source of phosphorus for zooplankton. Hydrobiologia 206: 217–223.
Hrbáček, J. & M. Hrbáčková-Esslová, 1960. Fish stock as a protective agent in the occurrence of slow-developing dwarf species and strains in the genus Daphnia. Int. Rev. ges. Hydrobiol. 45: 355–358.
Ketola, M. & I. Vuorinen, 1989. Modification of life-history parameters of Daphnia pulex Leydig and D. magna Straus by the presence of Chaoborus sp. Hydrobiologia 179: 149–155.
Kopáček, J. & J. Hejzlar, 1993. Semi-micro determination of total phosphorus in fresh waters with perchloric acid digestion. Internat. J. Envir. Anal. Chem. 53: 173–183.
Kubečka, J. & M. Wittingerová, 1998. Horizontal beaming as a crucial component of acoustic fish stock assessment in freshwater reservoirs. Fish. Res. 35: 99–106.
Lampert, W., 1987. Feeding and nutrition in Daphnia. Mem. Ist. ital. Idrobiol. 45: 143–192.
Lampert, W., 1993. Phenotypic plasticity of the size at first reproduction in Daphnia: the importance of maternal size. Ecology 74: 1455–1466.
Loose, C. J., E. Von Elert & P. Dawidowicz, 1993. Chemically induced diel vertical migration in Daphnia: a new bioassay for kairomones exuded by fish. Arch. Hydrobiol. 126: 329–337.
Lorentzen, C. J., 1967. Determination of chlorophyll and pheopigments: spectrophotometric equations. Limnol. Oceanogr. 12: 243–246.
Lüning, J., 1992. Phenotypic plasticity of Daphnia pulex in the presence of invertebrate predators: morphological and life history responses. Oecologia 92: 383–390.
Macháček, J., 1991. Indirect effect of planktivorous fish on the growth and reproduction of Daphnia galeata. Hydrobiologia 225: 193–197.
Macháček, J., 1993. Comparison of the response of Daphnia galeata and Daphnia obtusa to fish-produced chemical substance. Limnol. Oceanogr. 38: 1544–1550.
Macháček, J. & J. Sed'a, 1998. Spatio-temporal changes of morphological and life history parameters in Daphnia galeata in a canyon-shaped dam lake. Int. Rev. Hydrobiol. 83: 171–178.
Matveev, V., 1993. An investigation of allelopathic effects of Daphnia. Freshwat. Biol. 29: 99–105.
Monel, M.-L. & N. Lair, 1997. Relationships between heterotrophic nanoflagellates and the demographic response of Daphnia longispina in a eutrophic lake with poor food quality conditions. Freshwat. Biol. 38: 739–752.
Müller-Navarra, D. C., 1995. Biochemical versus mineral limitation in Daphnia. Limnol. Oceanogr. 40: 1209–1214.
Porter, K. G., 1973. Selective grazing and differential digestion of algae by zooplankton. Nature 244: 179–180.
Sanders, R. W., C. E. Williamson, P. L. Stutzman, R. E. Moeller, C. E. Goulden & R. Aoki-Goldsmith, 1996. Reproductive success of ‘herbivorous’ zooplankton fed algal and non-algal food resources. Limnol. Oceanogr. 41: 1295–1305.
Stibor, H., 1992. Predator induced life-history shifts in a freshwater cladoceran. Oecologia 92: 162–165.
Urabe, J., J. Clasen & R.W. Sterner, 1997. Phosphorus limitation of Daphnia growth: is it real? Limnol. Oceanogr. 42: 1436–1443.
Van Donk, E., M. Lürling, D. O. Hessen & G. M. Lokhorst, 1997. Altered cell wall morphology in nutrient-deficient phytoplankton and its impact on grazers. Limnol. Oceanogr. 42: 357–364.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Macháček, J. Daphnia galeata life history response to heterogeneous food conditions and dissolved chemicals in the Rímov Reservoir. Hydrobiologia 442, 215–222 (2001). https://doi.org/10.1023/A:1017524128909
Issue Date:
DOI: https://doi.org/10.1023/A:1017524128909