Abstract
Objectives
Class IV lupus nephritis (LN) is one of the most frequent and severe types of involvement in pediatric systemic lupus erythematosus. Gold standard treatment consists of intravenous (i.v.) Cyclophosphamide (CYC) associated with corticosteroids. Recent studies in adults have shown similar efficacy of oral Mycophenolate Mofetil (MMF) with fewer adverse events. Our aim was to compare the efficacy and tolerance of CYC and MMF as induction therapy in children with class IV LN.
Methods
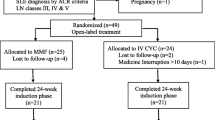
We conducted a retrospective study of children diagnosed with class IV LN who started oral MMF or i.v. CYC treatment at Necker Enfants Malades Hospital (Paris, France).
Results
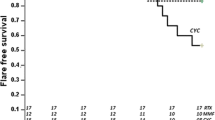
The study included 33 patients, 17 treated with oral MMF (51%) and 16 with i.v. CYC (48%). The characteristics at treatment induction did not significantly differ between the two groups except for the neurological involvement, that was only present in the CYC group. Complete remission was obtained in 9/17 (53%) children treated with MMF versus 10/16 (71%) treated with CYC (p = 0.46). Relapse was observed in 59% of patients receiving MMF versus 50% receiving CYC (p = 0.87), after a median of 3.4 years and 4.7 years after the beginning of treatment, respectively (p = 0.41). During the 6.5 years of follow-up, we observed no significant difference regarding the number of treatment-related adverse events between the two groups (p = 0.48).
Conclusion
We report similar efficacy and tolerance of MMF or CYC as induction therapy of class IV LN in children. However, the long-term adverse events such as infertility could not be systematically evaluated in this retrospective pediatric study. Overall, however, considering the long-term safety profile reported in the literature, we suggest that MMF may be used as first-line induction therapy in LN.
Graphical abstract



Similar content being viewed by others
Abbreviations
- ACR:
-
American College of Rheumatology
- CYC:
-
Cyclophosphamide
- eGFR:
-
Estimated glomerular filtration rate
- ISN/RPS:
-
International Society of Nephrology/Renal Pathology Society
- i.v.:
-
Intravenous
- LN:
-
Lupus nephritis
- MMF:
-
Mycophenolate mofetil
- NIH:
-
National Institutes of Health
- SAE:
-
Severe adverse events
- SHARE:
-
Single Hub and Access point for pediatric Rheumatology in Europe
- SLE:
-
Systemic lupus erythematosus
References
Groot N, de Graeff N, Marks SD, Brogan P, Avcin T, Bader-Meunier B et al (2017) European evidence-based recommendations for the diagnosis and treatment of childhood-onset lupus nephritis: the SHARE initiative. Ann Rheum Dis 76(12):1965–1973
Watson L, Leone V, Pilkington C, Tullus K, Rangaraj S, McDonagh JE et al (2012) Disease activity, severity, and damage in the UK Juvenile-Onset Systemic Lupus Erythematosus Cohort. Arthritis Rheum 64(7):2356–2365
Pinheiro SVB, Dias RF, Fabiano RCG, Araujo SA, Silva ACS (2019) Pediatric lupus nephritis. J Bras Nefrol 41(2):252–265
Therapy of lupus nephritis. Controlled trial of prednisone and cytotoxic drugs. - PubMed - NCBI. [cité 24 déc 2019]. Disponible sur: https://www.ncbi.nlm.nih.gov/pubmed/3511372
Houssiau FA, Vasconcelos C, D’Cruz D, Sebastiani GD, Garrido EER, Danieli MG et al (2002) Immunosuppressive therapy in lupus nephritis: the Euro-Lupus Nephritis Trial, a randomized trial of low-dose versus high-dose intravenous cyclophosphamide. Arthritis Rheum 46(8):2121–2131
Kingdon EJ, McLean AG, Psimenou E, Davenport A, Powis SH, Sweny P et al (2001) The safety and efficacy of MMF in lupus nephritis: a pilot study. Lupus 10(9):606–611
Ginzler E, Dooley M, Aranow C, Kim MY, Buyon J, Merrill J et al (2005) Mycophenolate mofetil or intravenous cyclophosphamide for lupus nephritis. N Engl J Med 353:2219–2228
Palmer SC, Tunnicliffe DJ, Singh-Grewal D, Mavridis D, Tonelli M, Johnson DW et al (2017) Induction and maintenance immunosuppression treatment of proliferative lupus nephritis: a network meta-analysis of randomized trials. Am J Kidney Dis sept 70(3):324–336
Mina R, von Scheven E, Ardoin SP, Eberhard BA, Punaro M, Ilowite N et al (2012) Consensus treatment plans for induction therapy of newly diagnosed proliferative lupus nephritis in juvenile systemic lupus erythematosus. Arthritis Care Res 64(3):375–383
Lau KK, Ault BH, Jones DP, Butani L (2008) Induction therapy for pediatric focal proliferative lupus nephritis: cyclophosphamide versus mycophenolate Mofetil. J Pediatr Health Care 22(5):282–288
Garcelon N, Neuraz A, Salomon R, Faour H, Benoit V, Delapalme A et al (2018) A clinician friendly data warehouse oriented toward narrative reports: Dr. Warehouse. J Biomed Inform avr 80:52–63
Hochberg MC (1997) Updating the American College of Rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 40(9):1725
Weening JJ, D’Agati VD, Schwartz MM, Seshan SV, Alpers CE, Appel GB et al (2004) The classification of glomerulonephritis in systemic lupus erythematosus revisited. Kidney Int 65(2):521–530
Tian SY, Feldman BM, Beyene J, Brown PE, Uleryk EM, Silverman ED (2014) Immunosuppressive therapies for the induction treatment of proliferative lupus nephritis: a systematic review and network metaanalysis. J Rheumatol 41(10):1998–2007
Appel GB, Contreras G, Dooley MA, Ginzler EM, Isenberg D, Jayne D et al (2009) Mycophenolate mofetil versus cyclophosphamide for induction treatment of lupus nephritis. J Am Soc Nephrol JASN 20(5):1103–1112
Smith EMD, Al-Abadi E, Armon K, Bailey K, Ciurtin C, Davidson J et al (2019) Outcomes following mycophenolate mofetil versus cyclophosphamide induction treatment for proliferative juvenile-onset lupus nephritis. Lupus. https://doi.org/10.1177/0961203319836712?url_ver=Z39.88-2003&rfr_id=ori%3Arid%3Acrossref.org&rfr_dat=cr_pub++0pubmed
Tamirou F, Husson SN, Gruson D, Debiève F, Lauwerys BR, Houssiau FA (2017) Brief report: The Euro-Lupus low-dose intravenous cyclophosphamide regimen does not impact the ovarian reserve, as measured by serum levels of anti-Müllerian Hormone. Arthritis Rheumatol Hoboken NJ 69(6):1267–1271
Funding
None.
Author information
Authors and Affiliations
Contributions
OB and BBM designed the study. LAE, MC, OB and BBM designed and created the clinical database. QR performed the statistical analysis. LAE, MC, OB and BMM analyzed and interpreted the data and drafted the article. All authors were involved in data collections, review and approval of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest to disclose. The authors have no financial relationship relevant to this article to disclose.
Ethical statement
The study was approved by the ethical committee of Necker-Enfants Malades hospital (APHP general register Number 2020 0805180729).
Clinical trial registration
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chbihi, M., Eveillard, LA., Riller, Q. et al. Induction therapy for pediatric onset class IV lupus nephritis: Mycophenolate Mofetil versus Cyclophosphamide. J Nephrol 36, 829–839 (2023). https://doi.org/10.1007/s40620-022-01438-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40620-022-01438-2