Abstract
Objectives
The primary purpose of this study was to determine whether both zoledronic acid (ZA) and denosumab (Dmab) equally suppress bone remodeling of the normal mandible, and the secondary purpose was to determine the influence of ZA and Dmab on other normal bones.
Methods
18F-sodium fluoride-positron-emission-tomography (18F-NaF-PET) was used to perform quantitative analysis of the bone metabolism in various parts. The end points of the study were the mean standardized uptake value (SUV) of each member of the ZA group (n = 9), the Dmab group (n = 16), and the Control group (n = 23).
Results
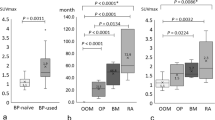
The SUV at the thoracic vertebrae in the ZA group were significantly lower than those of the Dmab and Control group (p < 0.05) In addition, the mean SUVs of the cervical vertebrae in the ZA group were significantly lower than those in the Control group (p < 0.05). There was no significant difference among ZA, Dmab and Control group in the other sites. There was no significant difference between the Dmab and Control groups at all sites.
Conclusions
The remodeling of mandible was not suppressed due to the treatment with anti-resorptive agents. Differences in the mechanisms of action between the BP and Dmab caused the specificity of the effect on the metabolism of normal bone.


Similar content being viewed by others
References
Coleman RE. Metastatic bone disease: clinical features, pathophysiology and treatment strategies. Cancer Treat Rev. 2001;27:165–76.
Lipton A. Management of bone metastases in breast cancer. Curr Treat Options Oncol. 2005;6:161–71.
Coleman RE, Rubens RD. The clinical course of bone metastases from breast cancer. Br J Cancer. 1987;55:61–6.
O’Reilly SM, Richards MA, Rubens RD. Liver metastases from breast cancer: the relationship between clinical, biochemical and pathological features and survival. Eur J Cancer (Oxford, England: 1990). 1990;26:574–7.
Wu S, Dahut WL, Gulley JL. The use of bisphosphonates in cancer patients. Acta Oncol. 2007;46:581–91.
McClung M, Harris ST, Miller PD, et al. Bisphosphonate therapy for osteoporosis: benefits, risks, and drug holiday. Am J Med. 2013;126:13–20.
Ruggiero SL, Dodson TB, Fantasia J, et al. American association of oral and maxillofacial surgeons position paper on medication-related osteonecrosis of the jaw - 2014 update. J Oral Maxillofac Surg. 2014;72:1938–56.
Khan AA, Morrison A, Hanley DA, et al. Diagnosis and management of osteonecrosis of the jaw: a systematic review and international consensus. J Bone Miner Res. 2015;30:3–23.
Takahashi M, Ozaki Y, Kizawa R, et al. Atypical femoral fracture in patients with bone metastasis receiving denosumab therapy: a retrospective study and systematic review. BMC Cancer. 2019;19:980.
Limones A, Sáez-Alcaide LM, Díaz-Parreño SA, et al. Medication-related osteonecrosis of the jaws (MRONJ) in cancer patients treated with denosumab VS. zoledronic acid: a systematic review and meta-analysis. Med Oral Patologia Oral y Cirugia Bucal. 2020;25:e326–36.
Lockwood M, Banderudrappagari R, Suva LJ, Makhoul I. Atypical femoral fractures from bisphosphonate in cancer patients—review. J Bone Oncol. 2019;18:100259.
Shane E, Burr D, Abrahamsen B, et al. Atypical subtrochanteric and diaphyseal femoral fractures: second report of a task force of the American Society for Bone and Mineral Research. J Bone Miner Res. 2014;29:1–23.
Gong X, Yu W, Zhao H, Su J, Sheng Q. Skeletal site-specific effects of zoledronate on in vivo bone remodeling and in vitro BMSCs Osteogenic Activity. Sci Rep. 2017;7:36129.
Chang J, Hakam AE, McCauley LK. Current understanding of the pathophysiology of osteonecrosis of the jaw. Curr Osteoporos Rep. 2018;16:584–95.
Blau M, Nagler W, Bender MA. Fluorine-18: a new isotope for bone scanning. J Nucl Med. 1962;3:332–4.
Li Y, Schiepers C, Lake R, Dadparvar S, Berenji GR. Clinical utility of 18F-fluoride PET/CT in benign and malignant bone diseases. Bone. 2012;50:128–39.
Even-Sapir E, Mishani E, Flusser G, Metser U. 18F-fluoride positron emission tomography and positron emission tomography/computed tomography. Semin Nucl Med. 2007;37:462–9.
Grant FD, Fahey FH, Packard AB, et al. Skeletal PET with 18F-fluoride: applying new technology to an old tracer. J Nucl Med. 2008;49:68–78.
Langsteger W, Rezaee A, Pirich C, Beheshti M. (18)F-NaF-PET/CT and (99m)Tc-MDP bone scintigraphy in the detection of bone metastases in prostate cancer. Semin Nucl Med. 2016;46:491–501.
Ristow O, Gerngroß C, Schwaiger M, et al. Effect of antiresorptive drugs on bony turnover in the jaw: denosumab compared with bisphosphonates. Br J Oral Maxillofac Surg. 2014;52:308–13.
Baron R, Ferrari S, Russell RGG. Denosumab and bisphosphonates: different mechanisms of action and effects. Bone. 2011;48:677–92.
Plotkin LI, Bivi N, Bellido T. A bisphosphonate that does not affect osteoclasts prevents osteoblast and osteocyte apoptosis and the loss of bone strength induced by glucocorticoids in mice. Bone. 2011;49:122–7.
Rosen LS, Gordon D, Tchekmedyian S, et al. Zoledronic acid versus placebo in the treatment of skeletal metastases in patients with lung cancer and other solid tumors: a phase III, double-blind, randomized trial—the Zoledronic Acid Lung Cancer and Other Solid Tumors Study Group. J Clin Oncol. 2003;21:3150–7.
Cole L, Vargo-Gogola T, Roeder R. Targeted delivery to bone and mineral deposits using bisphosphonate ligands. Adv Drug Deliv Rev. 2015;99:12.
Lin JH. Bisphosphonates: a review of their pharmacokinetic properties. Bone. 1996;18:75–85.
Manolagas SC. Birth and death of bone cells: basic regulatory mechanisms and implications for the pathogenesis and treatment of osteoporosis*. Endocr Rev. 2000;21:115–37.
Matsuura T, Mizumachi E, Katafuchi M, et al. Sex-related differences in cortical and trabecular bone quantities at the mandibular molar. J Hard Tissue Biol. 2014;23:267–74.
Blau M, Ganatra R, Bender MA. 18F-fluoride for bone imaging. Semin Nucl Med. 1972;2:31–7.
Cook GJ, Fogelman I. Detection of bone metastases in cancer patients by 18F-fluoride and 18F-fluorodeoxyglucose positron emission tomography. Q J Nucl Med. 2001;45:47–52.
Bortot DC, Amorim BJ, Oki GC, et al. 18F-Fluoride PET/CT is highly effective for excluding bone metastases even in patients with equivocal bone scintigraphy. Eur J Nucl Med Mol Imaging. 2012;39:1730–6.
Sarikaya I, Elgazzar AH, Sarikaya A, Alfeeli M. Normal bone and soft tissue distribution of fluorine-18-sodium fluoride and artifacts on 18F-NaF PET/CT bone scan: a pictorial review. Nucl Med Commun. 2017;38:810–9.
Win AZ, Aparici CM. Normal SUV values measured from NaF18- PET/CT bone scan studies. PLoS ONE. 2014;9:e108429-e.
Raje N, Woo S-B, Hande K, et al. Clinical, radiographic, and biochemical characterization of multiple myeloma patients with osteonecrosis of the jaw. Clin Cancer Res. 2008;14:2387–95.
Arce K, Assael LA, Weissman JL, Markiewicz MR. Imaging findings in bisphosphonate-related osteonecrosis of jaws. J Oral Maxillofac Surg. 2009;67:75–84.
Wilde F, Steinhoff K, Frerich B, et al. Positron-emission tomography imaging in the diagnosis of bisphosphonate-related osteonecrosis of the jaw. Oral Surg Oral Med Oral Pathol Oral Radiol Endodontol. 2009;107:412–9.
Funding
This study did not receive any funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee (the Kagawa University Ethical Committee (2020-#122)) and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fushimi, M., Ohbayashi, Y., Nakai, F. et al. The effect of zoledronic acid and denosumab on the mandible and other bones: a 18F-NaF-PET study. Oral Radiol 38, 594–600 (2022). https://doi.org/10.1007/s11282-022-00594-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11282-022-00594-2