Abstract
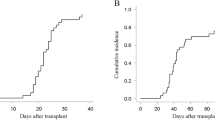
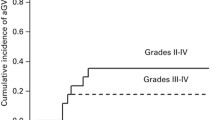
Adult T-cell leukemia/lymphoma (ATLL) is a peripheral T-cell lymphoma with a poor prognosis when treated with chemotherapy alone; therefore, allogeneic stem cell transplantation is a consideration. We attempted cord blood transplantation (CBT) using a reduced-intensity conditioning regimen without total body irradiation (non-TBI-RIC) to allow for the best possible timing of transplantation and improve survival outcomes, particularly in older patients. Forty-eight patients (27 male, 21 female) underwent CBT using fludarabine (Flu) 125 mg/m2 and melphalan (Mel) 140 mg/m2 as pre-transplant conditioning. The median age was 32 years (range 44–72), and 21 patients were in complete remission (CR) at the time of CBT. The median duration to neutrophil engraftment (NE) was 19.5 days (range 15–50), with a cumulative incidence of NE of 86.7% at day 50 after CBT. The 1- and 3-year overall survival (OS) rates were 40.4% and 37.7%, respectively. The 3-year OS rate in CR patients was 60.8%, compared with 18.8% in non-CR patients. In ATLL patients, CBT with non-TBI-RIC using Flu/Mel is a promising treatment strategy.






Similar content being viewed by others
Change history
17 March 2021
A Correction to this paper has been published: https://doi.org/10.1007/s12185-021-03125-7
References
Tsukasaki K, Utsunomiya A, Fukuda H, Shibata T, Fukushima T, Takatsuka Y, et al. VCAP-AMP-VECP compared with biweekly CHOP for adult t-cell leukemia-lymphoma: Japan Clinical Oncology Group Study JCOG9801. J Clin Oncol. 2007;25:5458–64.
Bazarbachi A, Plumelle Y, Ramos JC, Tortevoye P, Otrock Z, Taylor G, et al. Meta-analysis on the use of zidovudine and interferon-alfa in adult T-cell leukemia/lymphoma showing improved survival in the leukemic subtypes. J Clin Oncol. 2010;28:4177–83.
Katsuya H, Yamanaka T, Ishitsuka K, Utsunomiya A, Sasaki H, Hanada S, et al. Prognostic index for acute- and lymphoma-type adult T-cell leukemia/lymphoma. J Clin Oncol. 2012;30:1635–40.
Shimoyama M. Diagnostic criteria and classification of clinical subtypes of adult T-cell leukaemia-lymphoma. A report from the Lymphoma Study Group. Br J Haematol. 1991;79:428–37.
Nosaka K, Iwanaga M, Imaizumi Y, Ishitsuka K, Ishizawa K, Ishida Y, et al. Epidemiological and clinical features of adult T-cell leukemia-lymphoma in Japan, 2010–2011: a nationwide survey. Cancer Sci. 2017;108:2478–86.
Utsunomiya A, Miyazaki Y, Takatsuka Y, Hanada S, Uozumi K, Yashiki S, et al. Improved outcome of adult T cell leukemia/lymphoma with allogeneic hematopoietic stem cell transplantation. Bone Marrow Transplant. 2001;27:15–20.
Hishizawa M, Kanda J, Utsunomiya A, Taniguchi S, Eto T, Moriuchi Y, et al. Transplantation of allogeneic hematopoietic stem cells for adult T-cell leukemia: a nationwide retrospective study. Blood. 2010;116:1369–76.
Ishida T, Hishizawa M, Kato K, Tanosaki R, Fukuda T, Taniguchi S, et al. Allogeneic hematopoietic stem cell transplantation for adult T-cell leukemia-lymphoma with special emphasis on preconditioning regimen: a nationwide retrospective study. Blood. 2012;120:1734–41.
Bazarbachi A, Cwynarski K, Boumendil A, Finel H, Fields P, Raj K, et al. Outcome of patients with HTLV-1-associated adult T-cell leukemia/lymphoma after SCT: a retrospective study by the EBMT LWP. Bone Marrow Transplant. 2014;49:1266–8.
Kato K, Choi I, Wake A, Uike N, Taniguchi S, Moriuchi Y, et al. Treatment of patients with adult T cell leukemia/lymphoma with cord blood transplantation: a Japanese nationwide retrospective survey. Biol Blood Marrow Transplant. 2014;20:1968–74.
Nakamura T, Oku E, Nomura K, Morishige S, Takata Y, Seki R, et al. Unrelated cord blood transplantation for patients with adult T-cell leukemia/lymphoma: experience at a single institute. Int J Hematol. 2012;96:657–63.
Fukushima T, Itonaga H, Moriuchi Y, Yoshida S, Taguchi J, Imaizumi Y, et al. Feasibility of cord blood transplantation in chemosensitive adult T-cell leukemia/lymphoma: a retrospective analysis of the Nagasaki Transplantation Network. Int J Hematol. 2013;97:485–90.
Horwitz ME, Morris A, Gasparetto C, Sullivan K, Long G, Chute J, et al. Myeloablative intravenous busulfan/fludarabine conditioning does not facilitate reliable engraftment of dual umbilical cord blood grafts in adult recipients. Biol Blood Marrow Transplant. 2008;14:591–4.
Yamamoto H, Uchida N, Yuasa M, Kageyama K, Ota H, Kajiet D, et al. A novel reduced-toxicity myeloablative conditioning regimen using full-dose busulfan, fludarabine, and melphalan for single cord blood transplantation provides durable engraftment and remission in nonremission myeloid malignancies. Biol Blood Marrow Transplant. 2016;22:1844–50.
Shimoni A, Hardan I, Shem-Tov N, Rand A, Herscovici C, Yerushalmi R, et al. Comparison between two fludarabine-based reduced-intensity conditioning regimens before allogeneic hematopoietic stem-cell transplantation: fludarabine/melphalan is associated with higher incidence of acute graft-versus-host disease and non-relapse mortality and lower incidence of relapse than fludarabine/busulfan. Leukemia. 2007;21(10):2109–16.
Baron F, Labopin M, Peniket A, Jindra P, Afanasyev B, Sanz MA, et al. Reduced-intensity conditioning with fludarabine and busulfan versus fludarabine and melphalan for patients with acute myeloid leukemia: a report from the Acute Leukemia Working Party of the European Group for Blood and Marrow Transplantation. Cancer. 2015;121(7):1048–55.
Damlaj M, Alkhateeb HB, Hefazi M, Partain DK, Hashmi S, Gastineau DA, et al. Fludarabine-busulfan reduced-intensity conditioning in comparison with fludarabine-melphalan is associated with increased relapse risk in spite of pharmacokinetic dosing. Biol Blood Marrow Transplant. 2016;22(8):1431–9.
Eapen M, Brazauskas R, Hemmer M, Perez WS, Steinert P, Horowitz MM, et al. Hematopoietic cell transplant for acute myeloid leukemia and myelodysplastic syndrome: conditioning regimen intensity. Blood Adv. 2018;2(16):2095–103.
Kanda Y. Investigation of the freely available easy-to-use software “EZR” for medical statistics. Bone Marrow Transplant. 2013;48:452–8.
Horwitz S, O’Connor OA, Pro B, Illidge T, Fanale M, Advani R, et al. Brentuximab vedotin with chemotherapy for CD30-positive peripheral T-cell lymphoma (ECHELON-2): a global, double-blind, randomised, phase 3 trial. Lancet. 2019;19(393):229–40.
Fuji S, Inoue Y, Utsunomiya A, Moriuchi Y, Uchimaru K, Choi I, et al. Pretransplantation anti-CCR4 antibody mogamulizumab against adult T-cell leukemia/lymphoma is associated with significantly increased risks of severe and corticosteroid-refractory graft-versus-host disease, nonrelapse mortality, and overall mortality. J Clin Oncol. 2016;34:3426–33.
Acknowledgements
The authors thank all patients struggling with this difficult disease, as well as all nurses and other healthcare providers at our institution. This work was supported in part by the practical research programs of the Japan Agency for Medical Research and Development (AMED) under Grant number 20ck0106616h0001 and 20ck0106482h0002.
Funding
The authors have nothing to disclose.
Author information
Authors and Affiliations
Contributions
NN, YT, and AU designed the research and wrote the manuscript. NN performed statistical analysis. NN, YT, AK, Masahito T, Takayoshi M, TT, JO, ST, and YI performed CBT and treated the patients. NN, YT, AK, Masahito T, Takayoshi M, TT, JO, Mayumi T, Torahiko M, ST, YI, and AU wrote the draft of the manuscript. All the authors contributed to the final version.
Corresponding author
Ethics declarations
Conflict of interests
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
About this article
Cite this article
Nakano, N., Takatsuka, Y., Kubota, A. et al. Cord blood transplantation with a reduced-intensity conditioning regimen using fludarabine and melphalan for adult T-cell leukemia/lymphoma. Int J Hematol 113, 861–871 (2021). https://doi.org/10.1007/s12185-021-03102-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12185-021-03102-0