Abstract
Background
The G1 and G2 alleles of apolipoprotein L1 (APOL1) are common in the Black population and associated with increased risk of focal segmental glomerulosclerosis (FSGS). The molecular mechanisms linking APOL1 risk variants with FSGS are not clearly understood, and APOL1’s natural absence in laboratory animals makes studying its pathobiology challenging.
Methods
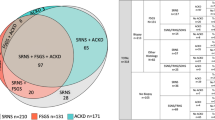
In a cohort of 90 Black patients with either FSGS or minimal change disease (MCD) enrolled in the Nephrotic Syndrome Study Network (58% pediatric onset), we used kidney biopsy traits as an intermediate outcome to help illuminate tissue-based consequences of APOL1 risk variants and expression. We tested associations between APOL1 risk alleles or glomerular APOL1 mRNA expression and 83 light- or electron-microscopy traits measuring structural and cellular kidney changes.
Results
Under both recessive and dominant models in the FSGS patient subgroup (61%), APOL1 risk variants were significantly correlated (defined as FDR <0.1) with decreased global mesangial hypercellularity, decreased condensation of cytoskeleton, and increased tubular microcysts. No significant correlations were detected in MCD cohort. Independent of risk alleles, glomerular APOL1 expression in FSGS patients was not correlated with morphologic features.
Conclusions
While APOL1-associated FSGS is associated with two risk alleles, both one and two risk alleles are associated with cellular/tissue changes in this study of FSGS patients. Our lack of discovery of a large group of tissue differences in FSGS and no significant difference in MCD may be due to the lack of power but also supports investigating whether machine learning methods may more sensitively detect APOL1-associated changes.




Similar content being viewed by others
Data Availability
Data are available upon request through communication with corresponding authors.
References
Genovese G, Friedman DJ, Ross MD, Lecordier L, Uzureau P, Freedman BI, Bowden DW, Langefeld CD, Oleksyk TK, Uscinski Knob AL, Bernhardy AJ, Hicks PJ, Nelson GW, Vanhollebeke B, Winkler CA, Kopp JB, Pays E, Pollak MR (2010) Association of trypanolytic ApoL1 variants with kidney disease in African Americans. Science 329:841–845
Tzur S, Rosset S, Shemer R, Yudkovsky G, Selig S, Tarekegn A, Bekele E, Bradman N, Wasser WG, Behar DM, Skorecki K (2010) Missense mutations in the APOL1 gene are highly associated with end stage kidney disease risk previously attributed to the MYH9 gene. Hum Genet 128:345–350
Kopp JB, Nelson GW, Sampath K, Johnson RC, Genovese G, An P, Friedman D, Briggs W, Dart R, Korbet S, Mokrzycki MH, Kimmel PL, Limou S, Ahuja TS, Berns JS, Fryc J, Simon EE, Smith MC, Trachtman H, Michel DM, Schelling JR, Vlahov D, Pollak M, Winkler CA (2011) APOL1 genetic variants in focal segmental glomerulosclerosis and HIV-associated nephropathy. J Am Soc Nephrol 22:2129–2137
Ng DK, Robertson CC, Woroniecki RP, Limou S, Gillies CE, Reidy KJ, Winkler CA, Hingorani S, Gibson KL, Hjorten R, Sethna CB, Kopp JB, Moxey-Mims M, Furth SL, Warady BA, Kretzler M, Sedor JR, Kaskel FJ, Sampson MG (2017) APOL1-associated glomerular disease among African-American children: a collaboration of the Chronic Kidney Disease in Children (CKiD) and Nephrotic Syndrome Study Network (NEPTUNE) cohorts. Nephrol Dial Transplant 32:983–990
Sampson MG, Robertson CC, Martini S, Mariani LH, Lemley KV, Gillies CE, Otto EA, Kopp JB, Randolph A, Vega-Warner V, Eichinger F, Nair V, Gipson DS, Cattran DC, Johnstone DB, O'Toole JF, Bagnasco SM, Song PX, Barisoni L, Troost JP, Kretzler M, Sedor JR, Nephrotic Syndrome Study Network (2016) Integrative genomics identifies novel associations with APOL1 risk genotypes in Black NEPTUNE subjects. J Am Soc Nephrol 27:814–823
Wen H, Kumar V, Lan X, Shoshtari SSM, Eng JM, Zhou X, Wang F, Wang H, Skorecki K, Xing G, Wu G, Luo H, Malhotra A, Singhal PC (2018) APOL1 risk variants cause podocytes injury through enhancing endoplasmic reticulum stress. Biosci Rep 38:BSR20171713
Nichols B, Jog P, Lee JH, Blackler D, Wilmot M, D'Agati V, Markowitz G, Kopp JB, Alper SL, Pollak MR, Friedman DJ (2015) Innate immunity pathways regulate the nephropathy gene Apolipoprotein L1. Kidney Int 87:332–342
Ma L, Chou JW, Snipes JA, Bharadwaj MS, Craddock AL, Cheng D, Weckerle A, Petrovic S, Hicks PJ, Hemal AK, Hawkins GA, Miller LD, Molina AJ, Langefeld CD, Murea M, Parks JS, Freedman BI (2017) APOL1 Renal-risk variants induce mitochondrial dysfunction. J Am Soc Nephrol 28:1093–1105
Beckerman P, Bi-Karchin J, Park AS, Qiu C, Dummer PD, Soomro I, Boustany-Kari CM, Pullen SS, Miner JH, Hu CA, Rohacs T, Inoue K, Ishibe S, Saleem MA, Palmer MB, Cuervo AM, Kopp JB, Susztak K (2017) Transgenic expression of human APOL1 risk variants in podocytes induces kidney disease in mice. Nat Med 23:429–438
Fu Y, Zhu JY, Richman A, Zhang Y, Xie X, Das JR, Li J, Ray PE, Han Z (2017) APOL1-G1 in nephrocytes induces hypertrophy and accelerates cell death. J Am Soc Nephrol 28:1106–1116
Ma L, Shelness GS, Snipes JA, Murea M, Antinozzi PA, Cheng D, Saleem MA, Satchell SC, Banas B, Mathieson PW, Kretzler M, Hemal AK, Rudel LL, Petrovic S, Weckerle A, Pollak MR, Ross MD, Parks JS, Freedman BI (2015) Localization of APOL1 protein and mRNA in the human kidney: nondiseased tissue, primary cells, and immortalized cell lines. J Am Soc Nephrol 26:339–348
Madhavan SM, O'Toole JF, Konieczkowski M, Ganesan S, Bruggeman LA, Sedor JR (2011) APOL1 localization in normal kidney and nondiabetic kidney disease. J Am Soc Nephrol 22:2119–2128
Kozlitina J, Zhou H, Brown PN, Rohm RJ, Pan Y, Ayanoglu G, Du X, Rimmer E, Reilly DF, Roddy TP, Cully DF, Vogt TF, Blom D, Hoek M (2016) Plasma levels of risk-variant APOL1 do not associate with renal disease in a population-based cohort. J Am Soc Nephrol 27:3204–3219
Kopp JB, Winkler CA, Zhao X, Radeva MK, Gassman JJ, D’ Agati VD, Nast CC, Wei C, Reiser J, Guay-Woodford LM, Pollak MR, Hildebrandt F, Moxey-Mims M, Gipson DS, Trachtman H, Friedman AL, Kaskel FJ, FSGS-CT Study Consortium (2015) Clinical features and histology of apolipoprotein L1-associated nephropathy in the FSGS clinical trial. J Am Soc Nephrol 26:1443–1448
Kopp JB, Nelson GW, Sampath K, Johnson RC, Genovese G, An P, Friedman D, Briggs W, Dart R, Korbet S, Mokrzycki MH, Kimmel PL, Limou S, Ahuja TS, Berns JS, Fryc J, Simon EE, Smith MC, Trachtman H, Michel DM, Schelling JR, Vlahov D, Pollak M, Winkler CA (2011) APOL1 genetic variants in focal segmental glomerulosclerosis and HIV-associated nephropathy. J Am Soc Nephrol 22:2129–2137
Larsen CP, Beggs ML, Saeed M, Walker PD (2013) Apolipoprotein L1 risk variants associate with systemic lupus erythematosus-associated collapsing glomerulopathy. J Am Soc Nephrol 24:722–725
Larsen CP, Beggs ML, Walker PD, Saeed M, Ambruzs JM, Messias NC (2014) Histopathologic effect of APOL1 risk alleles in PLA2R-associated membranous glomerulopathy. Am J Kidney Dis 64:161–163
Larsen CP, Beggs ML, Saeed M, Ambruzs JM, Cossey LN, Messias NC, Walker PD, Freedman BI (2015) Histopathologic findings associated with APOL1 risk variants in chronic kidney disease. Mod Pathol 28:95–102
Gadegbeku CA, Gipson DS, Holzman LB, Ojo AO, Song PX, Barisoni L, Sampson MG, Kopp JB, Lemley KV, Nelson PJ, Lienczewski CC, Adler SG, Appel GB, Cattran DC, Choi MJ, Contreras G, Dell KM, Fervenza FC, Gibson KL, Greenbaum LA, Hernandez JD, Hewitt SM, Hingorani SR, Hladunewich M, Hogan MC, Hogan SL, Kaskel FJ, Lieske JC, Meyers KE, Nachman PH, Nast CC, Neu AM, Reich HN, Sedor JR, Sethna CB, Trachtman H, Tuttle KR, Zhdanova O, Zilleruelo GE, Kretzler M (2013) Design of the Nephrotic Syndrome Study Network (NEPTUNE) to evaluate primary glomerular nephropathy by a multidisciplinary approach. Kidney Int 83:749–756
Barisoni L, Gimpel C, Kain R, Laurinavicius A, Bueno G, Zeng C, Liu Z, Schaefer F, Kretzler M, Holzman LB, Hewitt SM (2017) Digital pathology imaging as a novel platform for standardization and globalization of quantitative nephropathology. Clin Kidney J 10:176–187
Barisoni L, Nast CC, Jennette JC, Hodgin JB, Herzenberg AM, Lemley KV, Conway CM, Kopp JB, Kretzler M, Lienczewski C, Avila-Casado C, Bagnasco S, Sethi S, Tomaszewski J, Gasim AH, Hewitt SM (2013) Digital pathology evaluation in the multicenter Nephrotic Syndrome Study Network (NEPTUNE). Clin J Am Soc Nephrol 8:1449–1459
Ng DK, Schwartz GJ, Schneider MF, Furth SL, Warady BA (2018) Combination of pediatric and adult formulas yield valid glomerular filtration rate estimates in young adults with a history of pediatric chronic kidney disease. Kidney Int 94:170–177
Velez JCQ, Caza T, Larsen CP (2020) COVAN is the new HIVAN: the re-emergence of collapsing glomerulopathy with COVID-19. Nat Rev Nephrol 16:565–567
Olabisi OA, Zhang JY, VerPlank L, Zahler N, DiBartolo S 3rd, Heneghan JF, Schlondorff JS, Suh JH, Yan P, Alper SL, Friedman DJ, Pollak MR (2016) APOL1 kidney disease risk variants cause cytotoxicity by depleting cellular potassium and inducing stress-activated protein kinases. Proc Natl Acad Sci U S A 113:830–837
Lan X, Jhaveri A, Cheng K, Wen H, Saleem MA, Mathieson PW, Mikulak J, Aviram S, Malhotra A, Skorecki K, Singhal PC (2014) APOL1 risk variants enhance podocyte necrosis through compromising lysosomal membrane permeability. Am J Physiol Renal Physiol 307:F326–F336
Shah SS, Lannon H, Dias L, Zhang JY, Alper SL, Pollak MR, Friedman DJ (2019) APOL1 kidney risk variants induce cell death via mitochondrial translocation and opening of the mitochondrial permeability transition pore. J Am Soc Nephrol 30:2355–2368
Schaub C, Verdi J, Lee P, Terra N, Limon G, Raper J, Thomson R (2020) Cation channel conductance and pH gating of the innate immunity factor APOL1 is governed by pore lining residues within the C-terminal domain. J Biol Chem 295:13138–13149
Crews DC, Purnell TS (2020) COVID-19, Racism, and racial disparities in kidney disease: galvanizing the kidney community response. J Am Soc Nephrol 31:1–3
Kasembeli AN, Duarte R, Ramsay M, Mosiane P, Dickens C, Dix-Peek T, Limou S, Sezgin E, Nelson GW, Fogo AB, Goetsch S, Kopp JB, Winkler CA, Naicker S (2015) APOL1 risk variants are strongly associated with HIV-associated nephropathy in Black South Africans. J Am Soc Nephrol 26:2882–2890
O'Toole JF, Schilling W, Kunze D, Madhavan SM, Konieczkowski M, Gu Y, Luo L, Wu Z, Bruggeman LA, Sedor JR (2018) ApoL1 overexpression drives variant-independent cytotoxicity. J Am Soc Nephrol 29:869–879
Datta S, Kataria R, Zhang JY, Moore S, Petitpas K, Mohamed A, Zahler N, Pollak MR, Olabisi OA (2020) Kidney disease-asociated APOL1 variants have dose-dependent, dominant toxic gain-of-function. J Am Soc Nephrol 31:2083–2096
(1983) Childhood nephrotic syndrome associated with diffuse mesangial hypercellularity. A report of the Southwest Pediatric Nephrology Study Group. Kidney Int 24:87-94
Silverstein DM, Craver RD (2008) Mesangial hypercellularity in children: presenting features and outcomes. Pediatr Nephrol 23:921–928
Fujinaga S, Urushihara Y (2016) Favorable outcome in children with idiopathic steroid-resistant nephrotic syndrome due to mesangial hypercellularity: a distinct disease entity? Pediatr Nephrol 31:509–510
Wenderfer SE, Swinford RD, Braun MC (2010) C1q nephropathy in the pediatric population: pathology and pathogenesis. Pediatr Nephrol 25:1385–1396
Reiser J, Altintas MM (2016) Podocytes. F1000Res 5:F1000
Shi SF, Wang SX, Zhang YK, Zhao MH, Zou WZ (2008) Ultrastructural features and expression of cytoskeleton proteins of podocyte from patients with minimal change disease and focal segmental glomerulosclerosis. Ren Fail 30:477–483
Bruggeman LA, O'Toole JF, Sedor JR (2019) APOL1 polymorphisms and kidney disease: loss-of-function or gain-of-function? Am J Physiol Renal Physiol 316:F1–F8
Members of the Nephrotic Syndrome Study Network (NEPTUNE):
NEPTUNE Enrolling Centers
Cleveland Clinic, Cleveland, OH: K Dell*, J Sedor**, M Schachere#, J Negrey#
Children’s Hospital, Los Angeles, CA: K Lemley*, E Lim#
Children’s Mercy Hospital, Kansas City, MO: T Srivastava*, A Garrett#
Cohen Children’s Hospital, New Hyde Park, NY: C Sethna*, K Laurent #
Columbia University, New York, NY: P Canetta*, A Pradhan#
Emory University, Atlanta, GA: L Greenbaum*, C Wang**, C Kang#
Harbor-University of California Los Angeles Medical Center: S Adler*, J LaPage#
John H. Stroger Jr. Hospital of Cook County, Chicago, IL: A Athavale*, M Itteera
Johns Hopkins Medicine, Baltimore, MD: M Atkinson*, S Boynton#
Mayo Clinic, Rochester, MN: F Fervenza*, M Hogan**, J Lieske*, V Chernitskiy#
Montefiore Medical Center, Bronx, NY: F Kaskel*, M Ross*, P Flynn#
NIDDK Intramural, Bethesda MD: J Kopp*, J Blake#
New York University Medical Center, New York, NY: H Trachtman*, O Zhdanova**, F Modersitzki#, S Vento#
Stanford University, Stanford, CA: R Lafayette*, K Mehta#
Temple University, Philadelphia, PA: C Gadegbeku*, S Quinn-Boyle#
University Health Network Toronto: M Hladunewich**, H Reich**, P Ling#, M Romano#
University of Miami, Miami, FL: A Fornoni*, C Bidot#
University of Michigan, Ann Arbor, MI: M Kretzler*, D Gipson*, A Williams#, J LaVigne#
University of North Carolina, Chapel Hill, NC: V Derebail*, K Gibson*, E Cole#, J Ormond-Foster#
University of Pennsylvania, Philadelphia, PA: L Holzman*, K Meyers**, K Kallem#, A Swenson#
University of Texas Southwestern, Dallas, TX: K Sambandam*, Z Wang#, M Rogers#
University of Washington, Seattle, WA: A Jefferson*, S Hingorani**, K Tuttle**§, M Bray #, M Kelton#, A Cooper#§
Wake Forest University Baptist Health, Winston-Salem, NC: JJ Lin*, Stefanie Baker#
Data Analysis and Coordinating Center: M Kretzler, L Barisoni, J Bixler, H Desmond, S Eddy, D Fermin, C Gadegbeku, B Gillespie, D Gipson, L Holzman, V Kurtz, M Larkina, J Lavigne, S Li, S Li, CC Lienczewski, J Liu, T Mainieri, L Mariani, M Sampson, J Sedor, A Smith, A Williams, J Zee.
Digital Pathology Committee: Carmen Avila-Casado (University Health Network, Toronto), Serena Bagnasco (Johns Hopkins University), Joseph Gaut (Washington University in St Louis), Stephen Hewitt (National Cancer Institute), Jeff Hodgin (University of Michigan), Kevin Lemley (Children’s Hospital of Los Angeles), Laura Mariani (University of Michigan), Matthew Palmer (University of Pennsylvania), Avi Rosenberg (Johns Hopkins University), Virginie Royal (University of Montreal), David Thomas (University of Miami), Jarcy Zee (University of Pennsylvania) Co-Chairs: Laura Barisoni (Duke University) and Cynthia Nast (Cedar Sinai).
*Principal Investigator; **Co-investigator; #Study Coordinator
§Providence Medical Research Center, Spokane, WA
Funding
This study was supported by a grant from the National Institute of Diabetes, Digestive, and Kidney Diseases to MGS (NIDDK R01-DK108805). Support for JBK was provided by the NIDDK Intramural Research Program. The Nephrotic Syndrome Study Network Consortium (NEPTUNE), U54-DK-083912, is a part of the National Institutes of Health (NIH) Rare Disease Clinical Research Network (RDCRN), supported through a collaboration between the Office of Rare Diseases Research, National Center for Advancing Translational Sciences and the National Institute of Diabetes, Digestive, and Kidney Diseases. Additional funding and/or programmatic support for this project has also been provided by the University of Michigan, the NephCure Kidney International and the Halpin Foundation.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethics approval
The work presented here followed all ethical procedures.
Consent to participate/publication
All NEPTUNE participants consented to participate in the NEPTUNE studies and consented to have publications resulting from the use of their data.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zee, J., McNulty, M.T., Hodgin, J.B. et al. APOL1 genotype-associated morphologic changes among patients with focal segmental glomerulosclerosis. Pediatr Nephrol 36, 2747–2757 (2021). https://doi.org/10.1007/s00467-021-04990-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00467-021-04990-4