Abstract
Purpose
The clinical benefit of conversion surgery (CS) for unresectable gastric cancer (GC), whereby unresectable GC responds to chemotherapy and subsequently receives curative-intent surgery, remains unclear. Here, we aimed to clarify the clinical value of CS.
Methods
In this retrospective cohort study, we analyzed 175 unresectable GC, who received triple combined chemotherapy between 2004 and 2019. We divided patients into two groups: those who underwent CS and those receiving chemotherapy only (CS and C groups, respectively). Propensity score matching was used to minimize confounding bias.
Results
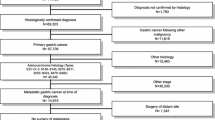
Of 175 cases, 61 (34.9%) underwent CS. R0 resection was obtained in 85.2%. After matching, 44 pairs were selected; there were no significant differences in baseline covariants. Group CS had a significantly better median overall survival (OS) (18.8 vs. 46.0 months, p < 0.001), and prolonged progression-free survival (7.4 vs. 25.8 months, p < 0.001). Subgroup analysis of OS showed a favorable trend for CS for almost all subgroups. Multivariate analysis revealed that good ECOG performance status and CS were associated with a longer OS.
Conclusion
The survival benefit of CS was consistently demonstrated in the univariate and multivariate analysis, even in the matched cohort. Additional large-scale trials are needed for further validation.



Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Beom SH, Choi YY, Baek SE, Li SX, Lim JS, Son T et al (2018) Multidisciplinary treatment for patients with stage IV gastric cancer: the role of conversion surgery following chemotherapy. BMC Cancer. https://doi.org/10.1186/s12885-018-4998-x
Bray F, Ferlay J, Soerjomataram I, Siegel R, Torre L, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68(6):394–424
Dindo D, Demartines N, Clavien PA (2004) Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg 240(2):205–213
Einama T, Abe H, Shichi S, Matsui H, Kanazawa R, Shibuya K et al (2017) Long-term survival and prognosis associated with conversion surgery in patients with metastatic gastric cancer. Mol Clin Oncol 6(2):163–166
Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45(2):228–247
Folprecht G, Grothey A, Alberts S, Raab HR, Köhne CH (2005) Neoadjuvant treatment of unresectable colorectal liver metastases: correlation between tumour response and resection rates. Ann Oncol 16(8):1311–1319
Fujitani K, Yang H, Mizusawa J, Kim Y, Terashima M, Han S et al (2016) Gastrectomy plus chemotherapy versus chemotherapy alone for advanced gastric cancer with a single non-curable factor (REGATTA): a phase 3, randomised controlled trial. Lancet Oncol 17(3):309–318
Fukuchi M, Ishiguro T, Ogata K, Suzuki O, Kumagai Y, Ishibashi K et al (2015) Prognostic role of conversion surgery for unresectable gastric cancer. Ann Surg Oncol 22(11):3618–3624
Han DS, Suh YS, Kong SH, Lee HJ, Im SA, Bang YJ et al (2013) Outcomes of surgery aiming at curative resection in good responder to induction chemotherapy for gastric cancer with distant metastases. J Surg Oncol 107(5):511–516
Japanese Gastric Cancer Association (2011) Japanese classification of gastric carcinoma: 3rd English edition. Gastric Cancer 14(2):101–112
Japanese Gastric Cancer Association (2020) Japanese gastric cancer treatment guidelines 2018 (5th edition). Gastric Cancer. https://doi.org/10.1007/s10120-020-01042-y
Jones RP, Hamann S, Malik HZ, Fenwick SW, Poston GJ, Folprecht G (2014) Defined criteria for resectability improves rates of secondary resection after systemic therapy for liver limited metastatic colorectal cancer. Eur J Cancer 50(9):1590–1601
Kanda T, Yajima K, Kosugi S, Ishikawa T, Ajioka Y, Hatakeyama K (2012) Gastrectomy as a secondary surgery for stage IV gastric cancer patients who underwent S-1-based chemotherapy: a multi-institute retrospective study. Gastric Cancer 15(3):235–244
Katayama H, Kurokawa Y, Nakamura K, Ito H, Kanemitsu Y, Masuda N et al (2016) Extended Clavien-Dindo classification of surgical complications: Japan Clinical Oncology Group postoperative complications criteria. Surg Today 46(6):668–685
Kim SW (2014) The result of conversion surgery in gastric cancer patients with peritoneal seeding. J Gastr Cancer 14(4):266–270
Kim S, Yoon M, Park Y, Kim M, Nam B, Park S (2018) Nomograms predicting survival of patients with unresectable or metastatic gastric cancer who receive combination cytotoxic chemotherapy as first-line treatment. Gastric Cancer 21(3):453–463
Kinoshita J, Fushida S, Tsukada T, Oyama K, Okamoto K, Makino I et al (2015) Efficacy of conversion gastrectomy following docetaxel, cisplatin, and S-1therapy in potentially resectable stage IV gastric cancer. Eur J Surg Oncol 41(10):1354–1360
Koo D, Ryoo B, Kim H, Ryu M, Lee S, Moon J et al (2011) A prognostic model in patients who receive chemotherapy for metastatic or recurrent gastric cancer: validation and comparison with previous models. Cancer Chemother Pharmacol 68(4):913–921
Maeda O, Matsuoka A, Miyahara R, Funasaka K, Hirooka Y, Fukaya M et al (2017) Modified docetaxel, cisplatin and capecitabine for stage IV gastric cancer in Japanese patients: a feasibility study. World J Gastroenterol 23(6):1090–1097
Mieno H, Yamashita K, Hosoda K, Moriya H, Higuchi K, Azuma M et al (2017) Conversion surgery after combination chemotherapy of docetaxel, cisplatin and S-1 (DCS) for far-advanced gastric cancer. Surg Today 47(10):1249–1258
Mitsui Y, Sato Y, Miyamoto H, Fujino Y, Takaoka T, Miyoshi J et al (2015) Trastuzumab in combination with docetaxel/cisplatin/S-1 (DCS) for patients with HER2-positive metastatic gastric cancer: feasibility and preliminary efficacy. Cancer Chemother Pharmacol 76(2):375–382
Morgagni P, Solaini L, Framarini M, Vittimberga G, Gardini A, Tringali D et al (2018) Conversion surgery for gastric cancer: a cohort study from a western center. Int J Surg 53:360–365
Muro K, Cutsem E, Narita Y, Pentheroudakis G, Baba E, Li J et al (2018) Pan-Asian adapted ESMO Clinical Practice Guidelines for the management of patients with metastatic gastric cancer: a JSMO–ESMO initiative endorsed by CSCO, KSMO, MOS. SSO and TOS Ann Oncol 30(1):19–33
Nakamura M, Ojima T, Nakamori M, Katsuda M, Tsuji T, Hayata K et al (2019) Conversion surgery for gastric cancer with peritoneal metastasis based on the diagnosis of second-look staging laparoscopy. J Gastroint Surg 23(9):1758–1766
National Cancer Institute (2009) National Cancer Institute Common Terminology Criteria for Adverse Events v 4.0. 2009. https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03/CTCAE_4.03_2010-06-14_QuickReference_5x7.pdf. Accessed 1 March 2020
National Comprehensive Cancer Network (2019) Gastric Cancer (version 1.2020). https://www.nccn.org/professionals/physician_gls/pdf/gastric.pdf. Accessed 1 March 2020
Ohnuma H, Sato Y, Hirakawa M, Kikuchi S, Miyanishi K, Sagawa T et al (2018) Docetaxel, cisplatin and S-1 (DCS) combination chemotherapy for gastric cancer patients with peritoneal metastasis: a retrospective study. Cancer Chemother Pharmacol 81(3):539–548
Saito M, Kiyozaki H, Takata O, Suzuki K, Rikiyama T (2014) Treatment of stageIV gastric cancer with induction chemotherapy using S-1 and cisplatin followed by curative resection in selected patients. World J Surg Oncol 12:406
Sakamoto Y, Sano T, Shimada K, Esaki M, Saka M, Fukagawa T et al (2007) Favorable indications for hepatectomy in patients with liver metastasis from gastric cancer. J Surg Oncol 95(7):534–539
Sasako M, Sano T, Yamamoto S, Kurokawa Y, Nashimoto A, Kurita A et al (2008) D2 lymphadenectomy alone or with para-aortic nodal dissection for gastric cancer. N Engl J Med 359(5):453–462
Sato Y, Takayama T, Sagawa T, Takahashi Y, Ohnuma H, Okubo Y et al (2010) Phase II study of S-1, docetaxel and cisplatin combination chemotherapy in patients with unresectable metastatic gastric cancer. Cancer Chemother Pharmacol 66(4):721–728
Sato S, Kunisaki C, Tanaka Y, Sato K, Miyamoto H, Yukawa N et al (2019a) Curative-intent surgery for stage IV advanced gastric cancer: who can undergo surgery and what are the prognostic factors for long-term survival? Ann Surg Oncol 26(13):4452–4463
Sato Y, Sagawa T, Ohnuma H, Hirakawa M, Takahashi Y, Hamaguchi K et al (2019b) A dose-escalation study of docetaxel, oxaliplatin, and S-1 (DOS) as a first-line therapy for patients with unresectable metastatic gastric cancer. Cancer Chemother Pharmacol 83(1):161–167
Satoh S, Okabe H, Teramukai S, Hasegawa S, Ozaki N, Ueda S et al (2012) Phase II trial of combined treatment consisting of preoperative S-1 plus cisplatin followed by gastrectomy and postoperative S-1 for stage IV gastric cancer. Gastric Cancer 15(1):61–69
Shirasu H, Tsushima T, Kawahira M, Kawai S, Kawakami T, Kito Y et al (2018) Role of hepatectomy in gastric cancer with multiple liver-limited metastases. Gastric Cancer 21(2):338–344
Sobin LGM, Wittekind C (2009) TNM classification of malignant tumours, 7th edn. Wiley-Blackwell, Hoboken
Solaini L, Ministrini S, Bencivenga M, D’Ignazio A, Marino E, Cipollari C et al (2019) Conversion gastrectomy for stage IV unresectable gastric cancer: a GIRCG retrospective cohort study. Gastric Cancer 22(6):1285–1293
Takayama T, Sato Y, Sagawa T, Okamoto T, Nagashima H, Takahashi Y et al (2007) Phase I study of S-1, docetaxel and cisplatin combination chemotherapy in patients with unresectable metastatic gastric cancer. Br J Cancer 97(7):851–856
Terashima M, Iwasaki Y, Mizusawa J, Katayama H, Nakamura K, Katai H et al (2019) Randomized phase III trial of gastrectomy with or without neoadjuvant S-1 plus cisplatin for type 4 or large type 3 gastric cancer, the short-term safety and surgical results: Japan Clinical Oncology Group Study (JCOG0501). Gastric Cancer 22(5):1044–1052
Tomasello G, Petrelli F, Ghidini M, Pezzica E, Passalacqua R, Steccanella F et al (2017) Tumor regression grade and survival after neoadjuvant treatment in gastro-esophageal cancer: a meta-analysis of 17 published studies. Eur J Surg Oncol 43(9):1607–1616
Uemura N, Kikuchi S, Sato Y, Ohnuma H, Okamoto K, Miyamoto H et al (2017) A phase II study of modified docetaxel, cisplatin, and S-1 (mDCS) chemotherapy for unresectable advanced gastric cancer. Cancer Chemother Pharmacol 80(4):707–713
Wan T, Zhang X, Liang C, Liao C, Li J, Zhou Y (2019) The prognostic value of a pathologic complete response after neoadjuvant therapy for digestive cancer: systematic review and meta-analysis of 21 studies. Ann Surg Oncol 26(5):1412–1420
Yamada Y, Boku N, Mizusawa J, Iwasa S, Kadowaki S, Nakayama N et al (2019) Docetaxel plus cisplatin and S-1 versus cisplatin and S-1 in patients with advanced gastric cancer (JCOG1013): an open-label, phase 3, randomised controlled trial. Lancet Gastroenterol Hepatol 4(7):501–510
Yamaguchi K, Yoshida K, Tanahashi T, Takahashi T, Matsuhashi N, Tanaka Y et al (2018) The long-term survival of stage IV gastric cancer patients with conversion therapy. Gastric Cancer 21(2):315–323
Ychou M, Boige V, Pignon JP, Conroy T, Bouché O, Lebreton G et al (2011) Perioperative chemotherapy compared with surgery alone for resectable gastroesophageal adenocarcinoma: an FNCLCC and FFCD multicenter phase III trial. J Clin Oncol 29(13):1715–1721
Yoshida K, Yamaguchi K, Okumura N, Tanahashi T, Kodera Y (2016) Is conversion therapy possible in stage IV gastric cancer: the proposal of new biological categories of classification. Gastric Cancer 19(2):329–338
Yoshida K, Kodera Y, Kochi M, Ichikawa W, Kakeji Y, Sano T et al (2019) Addition of docetaxel to oral fluoropyrimidine improves efficacy in patients with stage III gastric cancer: interim analysis of JACCRO GC-07, a randomized controlled trial. J Clin Oncol 37(15):1296–1304
Funding
This study did not receive any specific grant from funding agencies in the public, commercial, or not‐for‐profit sectors.
Author information
Authors and Affiliations
Contributions
Conceptualization and design, HO and YS; data acquisition, HO, YS, NO, KH, NH, MS, TM, TI, and TN; investigation, KM, KT, and KM; interpretation of data and results, HO and YS; writing and editing, HO; supervision, IT and JK. All authors read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interests
J. Kato received research funding from SymBio Pharmaceuticals, Alexion Pharmaceuticals, Ono Pharmaceutical, Asahi Kasei Pharma, Astellas Pharma, Eisai, Otsuka Pharmaceutical, Kyowa Hakko Kirin, Shionogi, Daiichi Sankyo, Taiho Pharmaceutical, Chugai Pharmaceutical, Toyama Chemical, and Pfizer Japan, outside the submitted work. The other authors have no conflict of interest.
Ethics approval and consent to participate
The ethical committee of Sapporo Medical University Hospital approved this study. In this retrospective study, patients’ consent was obtained by opt-out manner. Participating patients were excluded when they specified that they were unwilling to participate.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ohnuma, H., Sato, Y., Onoyama, N. et al. Survival benefit of conversion surgery after intensive chemotherapy for unresectable metastatic gastric cancer: a propensity score-matching analysis. J Cancer Res Clin Oncol 147, 2385–2396 (2021). https://doi.org/10.1007/s00432-021-03516-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-021-03516-7