Abstract
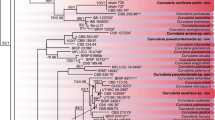
Colletotrichum spp. cause anthracnose disease in several plant species in tropical and temperate regions. In this study, a total of 30 isolates of Colletotrichum spp. were isolated from nine different fruit crops showing anthracnose symptoms. Phylogenetic analysis based on the GAPDH gene divided the population into four primary clades. All C. gloeosporioides species complex grouped together and separated from C. karstii and two different monophyletic lineages i.e. C. brevisporum, C. cliviae. C. gloeosporioides species complex was found dominant within the population containing 7 cryptic species. To study the population diversity of Colletotrichum species, the isolates were grouped into 4 geographical clusters based on their collection site i.e. Delhi, Maharashtra, West Bengal (WB), and North East (NE) states (Assam and Meghalaya). Out of 12 ISSR primers used 11 primers generated scorable polymorphic band of 100-1300 bp having average PIC value of 0.27. The intrapopulation genetic diversity was high in NE population with 78.75% polymorphism. The inter-population genetic distance was high (0.129) between Maharashtra and WB populations. The degree of genetic differentiation was 0.09 and gene flow was estimated at 2.45 within Colletotrichum population. Population stratification using UPGMA dendrogram divided the population into two main groups. Group 1 consisted of isolates from all over the four geographical region and population from NE region formed group 2. The overall result indicates admixture between isolates and homogeneous population structures from different geographical regions.




Similar content being viewed by others
References
Abang MM, Asiedu R, Hoffmann P, Wolf GA, Mignouna HD, Winter S (2006) Pathogenic and genetic variability among Colletotrichum gloeosporioides isolates from different yam hosts in the agroecological zones in Nigeria. J Phytopathol 154:51–61
Bezerra JP, Ferreira PV, da Barbosa LF, Ramos-Sobrinho R, Pinho DB, Reis A, Assuncao IP, Lima GSA (2016) First report of anthracnose on chayote fruits (Sechium edule) caused by Colletotrichum brevisporum. Plant Dis 100:217
Crous PW, Verkleij GJM, Groenewald JZ, Samson RA (2009) Fungal biodiversity. In: CBS Laboratory manual series no. 1. Centraalbureau voor Schimmelcultures, Utrecht
Dean R, Van Kan JAL, Pretorius ZA, Hammond-Kosack KE, Di Pietro A, Pietro DS, Rudd JJ, Dickman M, Kahmann R, Ellis J, Foster GD (2012) The Top 10 fungal pathogens in molecular plant pathology. Mol Plant Pathol 13:414–430
Farr DF, Aime MC, Rossman AY, Palm ME (2006) Species of Colletotrichum on agavaceae. Mycol Res 110:1395–1408
Freeman S, Katan T, Shabi E (1998) Characterization of Colletotrichum species responsible for anthracnose diseases of various fruits. Plant Dis 82(6):596–605
Freeman S, Horowitz-Brown S, Afanador-Kafuri L, Maymon M, Minz D (2013) Colletotrichum: host specificity and pathogenicity on selected tropical and subtropical crops. Acta Hort 975:209–216
Freeman S, Minz D, Maymon M, Zveibil A (2001) Genetic diversity within Colletotrichum acutatum sensu lato Simmonds. Phytopathol 91(6):586–592
Guarnaccia V, Groenewald JZ, Polizzi G, Crous PW (2017) High species diversity in Colletotrichum associated with citrus diseases in Europe. Persoonia 39:32–50
Guerber JC, Liu B, Correll JC, Johnston PR (2003) Characterization of diversity in Colletotrichum acutatum sensu lato by sequence analysis of two gene introns, mtDNA and intron RFLPs, and mating compatibility. Mycologia 95:872–895
Huang F, Chen GQ, Hou X, Fu YS, Cai L, Hyde KD, Li HY (2013) Colletotrichum species associated with cultivated citrus in China. Fungal Divers 61:61–74
Hyde KD, Nilsson RH, Alias SA et al (2009) Colletotrichum—names in current use. Fungal Divers 39:147–182
Irwin DE (2002) Phylogeographic breaks without geographic barriers to geneflow. Evol 56:2383–2394
Ivic D, Voncina D, Sever Z, Simon S, Pejic I (2013) Identification of Colletotrichum species causing bitter rot of apple and pear in Croatia. J Phytopathol 161:284–286
Jayasinghe CK, Fernando THPS (2009) First report of Colletotrichum acutatum on Mangifera indica in Sri Lanka. Ceylon J Sci (Biol Sci) 38:31–34
Jayawardena RS, Huang JK, Jin BC, Yan JY, Li XH, Hyde KD, Bahkali AH, Yin SL, Zhang GZ (2016a) An account of Colletotrichum species associated with strawberry anthracnose in China based on morphology and molecular data. Mycosphere 7:1147–1163
Jayawardena RS, Hyde KD, Damm U, Cai L, Liu M, Li XH, Zhang W, Zhao WS, Yan JY (2016b) Notes on currently accepted species of Colletotrichum. Mycosphere 7:1192–1260
Joa JH, Lim CK, Choi IY, Park MJ, Shin HD (2016) First report of Colletotrichum fructicola causing anthracnose on mango in Korea. Plant Dis 100:1793
Kim WG, Hong SK (2008) Occurrence of anthracnose on peach tree caused by Colletotrichum species. Plant Pathol J 24:80–83
Krishnapillai N, Wilson Wijeratnam RS (2014) First Report of Colletotrichum asianum causing anthracnose on Willard mangoes in Sri Lanka. N Dis Rep 29:1
Kumar N, Jhang T, Vir S (2010) Sharma TR (2010) Molecular and pathological characterization of Colletotrichum falcatum infecting subtropical Indian sugarcane. J Phytopathol 159:260–267
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874
Lima JS, Figueiredo JG, Gomes RG, Stringari D, Goulin EH, Adamoski D et al (2012) Genetic diversity of Colletotrichum spp. An endophytic fungi in a medicinal plant, Brazilian pepper tree. ISRN Microbiol. https://doi.org/10.5402/2012/215716
Lima NB, de Batista MVA, Morais MA Jr, De Barbosa MAG, Michereff SJ, Hyde KD, Camara MPS (2013) Five Colletotrichum species are responsible for mango anthracnose in north-eastern Brazil. Fungal Divers 61:75–88
Liu F, Wang M, Damm U, Crous PW, Cai L (2016) Species boundaries in plant pathogenic fungi: a Colletotrichum case study. BMC Evol Biol 16:1–14
Lu G, Cannon PF, Reid A, Simmons CM (2004) Diversity and molecular relationships of endophytic Colletotrichum isolates from the Iwokrama Forest Reserve, Guyana. Mycol Res 108:53–63
Marulanda ML, Lopez AM, Isaza L, Lopez P (2014) Microsatellite isolation and characterization for Colletotrichum spp, causal agent of anthracnose in Andean blackberry. Genet Mol Res 13:7673–7685
McDermott JM, McDonald BA (1993) Gene flow in plant pathosystems. Annu Rev Phytopathol 31:353–373
McDonald BA (1997) The population genetics of fungi: tools and techniques. Phytopathol 87:448–453
McDonald BA, Linda C (2002) Pathogen population genetics, evolutionary potential, and durable resistance. Annu Rev Phytopathol 40:349–379
McDonald BA, McDermott JM, Goodwin SB (1989) The population biology of host pathogen interactions. Annu Rev Phytopathol 27:77–94
McKay SF, Freeman S, Minz D, Maymon M, Sedgley M, Collins GC, Scott ES (2009) Morphological, genetic, and pathogenic characterization of Colletotrichum acutatum, the cause of anthracnose of almond in Australia. Phytopathol 99:985–995
Milgroom MG (1996) Recombination of multilocus structure of fungal populations. Annu Rev Phytopathol 34:457–477
Milgroom MG, Peever TL (2003) Population biology of plant pathogens; the synthesis of plant disease epidemiology and population genetics. Plant Dis 87:608–617
Moral J, Xavier C, Roca LF, Romero J, Moreda W, Trapero A (2014) La Antracnosis del olivo y su efecto en la calidad del aceite. Grasas Aceites 65:e028
Nascimento RJ, Mizubuti ESG, Câmara MPS, Ferreira MF, Maymon M, Freeman S, Michereff SJ (2010) First report of papaya fruit rot caused by Colletotrichum magna in Brazil. Plant Dis 94:1506
O’Connell RJ, Thon MR, Hacquard S, Amyotte SG, Kleemann J, Torres MF et al (2012) Lifestyle transitions in plant pathogenic Colletotrichum fungi deciphered by genome and transcriptome analyses. Nat Genet. https://doi.org/10.1038/ng.2372
Peakall PR, Smouse PE (2012) GenAlEx 6.5: genetic analysis in Excel. Population genetic software for teaching and research—an update. Bioinformatics 28:2537–2539
Phoulivong S, Cai L, Chen H, McKenzie EHC, Abdelsalam K, Chukeatirote E, Hyde KD (2010) Colletotrichum gloeosporioides is not a common pathogen on tropical fruits. Fungal Divers 44:33–43
Rampersad SN, Perez-Brito D, Torres-Calzada C, Tapia-Tussell R, Carrington CVF (2013) Genetic structure of Colletotrichum gloeosporioides sensu lato isolates infecting papaya inferred by multilocus ISSR markers. Phytopathol 103:182–189
Ratanacherdchai K, Wang HK, Lin FC, Soytong K (2010) ISSR for comparison of cross-inoculation potential of Colletotrichum capsici causing chilli anthracnose. Afri J Microbiol Res 4:76–83
Rojas EI, Rehner SA, Samuels GJ (2010) Colletotrichum gloeosporioides s. l. associated with Theobroma cacao and other plants in Panama: multilocus phylogenies distinguish host-associated pathogens from asymptomatic endophytes. Mycol 102:1318–1338
Saini TJ, Gupta SG, Anandalakshmi R (2016) First report of papaya anthracnose caused by Colletotrichum fructicola in India. New Dis Rep 34:27
Sharma G, Kumar N, Weir BS, Hyde KD, Shenoy BD (2013) Apmat gene marker can resolve Colletotrichum species: a case study with Mangifera indica. Fungal Divers 61:117–138
Sharma G, Pinnaka AK, Shenoy BD (2014) Infra-specific diversity of Colletotrichum truncatum associated with chilli anthracnose in India based on microsatellite marker analysis. Arch. Phytopathol. Plant Prot. 47:2509–2523
Sharma G, Marcel M, Stanley F (2017) Epidemiology, pathology and identification of Colletotrichum including a novel species associated with avocado (Persea americana) anthracnose in Israel. Sci Rep 7:15839
Sharma G, Shenoy BD (2013a) Colletotrichum fructicola and C. siamense are involved in chilli anthracnose in India. Arch Phytopathol Plant Protect 47:1179–1194
Sharma G, Shenoy BD (2013b) Multigene sequence-based identification of Colletotrichum cymbidiicola, C. karstii and C. phyllanthi from India. Czech Mycol 65:79–88
Sneath PHA, Sokal RR (1973) Numerical taxonomy. WH Freeman and Company, San Francisco
Souza A De, Delphino Carboniabc RC, Wickertd E, de Macedo Lemosab EG, de Goes A (2013) Lack of host specificity of Colletotrichum spp. isolates associated with anthracnose symptoms on mango in Brazil. Plant Pathol 62:1038–1047
Talbot NJ (2001) Nucleic acid isolation and analysis. In: Talbot NJ (ed) Molecular and cellular biology of filamentous fungi. Oxford University Press, Oxford, pp 23–26
Taylor JW, Jacobson DJ, Fisher MS (1999) The evolution of asexual fungi: reproduction, speciation and classification. Annu Rev Phytopathol 37:197–246
Velho A, Alaniz S, Casanova L, Mondino P, Stadnik M (2015) New insight into the characterization of Colletotrichum species associated with apple diseases in southern Brazil and Uruguay. Fungal Biol 119:229–244
Vieira WAS, Bezerra PA, Silva AC, Veloso JS, Camara MPS, Doyle VP (2019) Optimal markers for the identification of Colletotrichum species. https://doi.org/10.1101/659177
Weeds PL, Chakraborty S, Fernandes CD, d’A Charchar MJ, Ramesh CR, Kexian Y, Kelemu S (2003) Genetic diversity in Colletotrichum gloeosporioides from Stylosanthes spp. at centers of origin and utilization. Phytopathol 93:176–185
Weir BS, Johnston PR, Damm U (2012) The Colletotrichum gloeosporioides species complex. Stud Mycol 73:115–180
Yan JY, Jayawardena MMRS, Goonasekara ID, Wang Y, Zhang W, Liu M, Huang JB, Wang ZY, Shang JJ, Peng YL, Bahkali A, Hyde KD, Li XH (2015) Diverse species of Colletotrichum associated with grapevine anthracnose in China. Fungal Divers 71:233–246
Yang YL, Liu ZY, Cai L, Hyde KD, Yu ZN, McKenzie EHC (2009) Colletotrichum anthracnose of Amaryllidaceae. Fungal Divers 39:123–146
Yeh FC, Boyle T, Rongcai Y, Ye Z, Xiyan JM (1999) POPGENE Version 1.31. University of Alberta
Acknowledgements
We acknowledge the support received from ICAR-IARI, New Delhi-110012, India.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Das, A., Roy, B., Jangra, S. et al. Analysis of genetic diversity of Colletotrichum population causing anthracnose in fruit crops using ISSR markers. Indian Phytopathology 74, 69–80 (2021). https://doi.org/10.1007/s42360-020-00295-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42360-020-00295-y