Abstract
Background
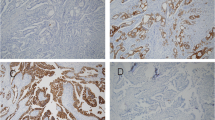
The incidence of adenocarcinoma of the ampulla of Vater has been increasing over the past years. Nevertheless, it is still a rare disease and the prognostic factors predicting long-term survival are not sufficiently clarified. This study aims to evaluate the association between histopathological characteristics and long-term survival of patients with ampullary cancer after curative resection, as well as the efficiency of immunohistochemical expression of CK7, CK20, and CDX2 to distinguish the histopathological (intestinal or pancreaticobiliary) patterns.
Methods
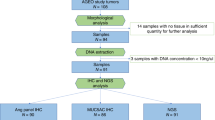
Demographic, histopathological data, pTNM stage, and immunohistochemical expression patterns were collected from 65 patients with adenocarcinoma of the ampulla of Vater. Five and 10-year overall and disease-free survival rates after curative resection were determined.
Results
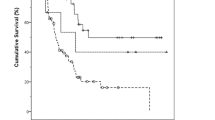
Of the 65 patients with ampullary carcinoma, 47 (72%) underwent radical resection. The 5- and 10-year overall survival rate was 46% and 37%, respectively. Our results demonstrate that the main prognostic factors were the presence and number of lymph node metastases, lymph node ratio (LNR), differentiation grade, and lymphovascular invasion. After multivariate analysis, only lymph node ratio ≥ 20% remained an independent prognostic factor of survival (HR: 2.63 95% CI: 1.05–6.61; p = 0.039).
Conclusion
Here, we demonstrated more evidence that the lymph node metastases are associated with poor prognosis in ampullary carcinoma. Particularly, the relation between the number of metastatic lymph nodes and the number of harvested lymph node (LNR) should be considered a major prognostic factor.



Similar content being viewed by others
References
Albores-Saavedra J, Schwartz AM, Batich K, Henson DE. Cancers of the ampulla of vater: demographics, morphology, and survival based on 5,625 cases from the SEER program. J Surg Oncol. 2009;100(7):598–605.
Winter JM, Cameron JL, Olino K, Herman JM, de Jong MC, Hruban RH, et al. Clinicopathologic analysis of ampullary neoplasms in 450 patients: implications for surgical strategy and long-term prognosis. J Gastrointest Surg. 2010;14(2):379–87.
Yamaguchi K, Tanaka M. Surgical treatment of carcinoma of the ampulla of Vater. In: Beger HG, Matsuno S, Cameron JL, editors. Diseases of the pancreas: current surgical therapy. Berlim: Springer-Verlag Berlin Heidelberg; 2008. p. 797–806.
Zhou H, Schaefer N, Wolff M, Fischer HP. Carcinoma of the ampulla of Vater: comparative histologic/immunohistochemical classification and follow-up. Am J Surg Pathol. 2004;28(7):875–82.
Riall TS, Cameron JL, Lillemoe KD, Winter JM, Campbell KA, Hruban RH, et al. Resected periampullary adenocarcinoma: 5-year survivors and their 6- to 10-year follow-up. Surgery. 2006;140(5):764–72.
Albores-Saavedra J, Henson DE, Klimstra DS. Malignant epithelial tumors of the ampulla. In: Rosai J, Sobin LH, editors. Tumors of the gallbladder, extrahepatic bile ducts, and ampulla of Vater: atlas of tumor pathology, third series. Washington, DC: Published by the Armed Forces Institute of Pathology under the auspices of Universities Associated for Research and Education in Pathology; 2000. p. 259–316.
Hsu HP, Yang TM, Hsieh YH, Shan YS, Lin PW. Predictors for patterns of failure after pancreaticoduodenectomy in ampullary cancer. Ann Surg Oncol. 2007;14(1):50–60.
Ha HR, Oh DY, Kim TY, Lee K, Kim K, Lee KH, et al. Survival outcomes according to adjuvant treatment and prognostic factors including host immune markers in patients with curatively resected ampulla of Vater cancer. PLoS One. 2016;11(3):e0151406.
Li Q, Liang L, Jia H, Li X, Xu Y, Zhu J, et al. Negative to positive lymph node ratio is a superior predictor than traditional lymph node status in stage III colorectal cancer. Oncotarget. 2016;7(44):72290–9.
Kim SH, Hwang HK, Lee WJ, Kang CM. Identification of an N staging system that predicts oncologic outcome in resected left-sided pancreatic cancer. Medicine (Baltimore). 2016;95(26):e4035.
Zhao LY, Li CC, Jia LY, Chen XL, Zhang WH, Chen XZ, et al. Superiority of lymph node ratio-based staging system for prognostic prediction in 2575 patients with gastric cancer: validation analysis in a large single center. Oncotarget. 2016;7(32):51069–81.
Shao Y, Geng Y, Gu W, Ning Z, Huang J, Pei H, et al. Assessment of lymph node ratio to replace the pN categories system of classification of the TNM system in esophageal squamous cell carcinoma. J Thorac Oncol. 2016;11(10):1774–84.
Kimura W, Futakawa N, Yamagata S, Wada Y, Kuroda A, Muto T, et al. Different clinicopathologic findings in two histologic types of carcinoma of papilla of Vater. Jpn J Cancer Res. 1994;85(2):161–6.
Kawabata Y, Tanaka T, Nishisaka T, Inao T, Nishi T, Yano S. Cytokeratin 20 (CK20) and apomucin 1 (MUC1) expression in ampullary carcinoma: correlation with tumor progression and prognosis. Diagn Pathol. 2010;5:75.
Chu PG, Schwarz RE, Lau SK, Yen Y, Weiss LM. Immunohistochemical staining in the diagnosis of pancreatobiliary and ampulla of Vater adenocarcinoma: application of CDX2, CK17, MUC1, and MUC2. Am J Surg Pathol. 2005;29(3):359–67.
Duval JV, Savas L, Banner BF. Expression of cytokeratins 7 and 20 in carcinomas of the extrahepatic biliary tract, pancreas, and gallbladder. Arch Pathol Lab Med. 2000;124(8):1196–200.
Hansel DE, Maitra A, Lin JW, Goggins M, Argani P, Yeo CJ, et al. Expression of the caudal-type homeodomain transcription factors CDX 1/2 and outcome in carcinomas of the ampulla of Vater. J Clin Oncol. 2005;23(9):1811–8.
Moskaluk CA, Zhang H, Powell SM, Cerilli LA, Hampton GM, Frierson HF Jr. Cdx2 protein expression in normal and malignant human tissues: an immunohistochemical survey using tissue microarrays. Modern Pathol. 2003;16(9):913–9.
Yun SP, Seo HI. Prognostic impact of immunohistochemical expression of CK7 and CK20 in curatively resected ampulla of Vater cancer. BMC Gastroenterol. 2015;15:165.
Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, et al. The eighth edition AJCC Cancer staging manual: continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017;67(2):93–9.
Kim SJ, An S, Kang HJ, Kim JY, Jang MA, Lee JH, et al. Validation of the eighth edition of the American Joint Committee on cancer staging system for ampulla of Vater cancer. Surgery. 2018;163(5):1071–9.
Hsu CH, Chen TD, Tsai CY, Hsu JT, Yeh CN, Jan YY, et al. Prognostic value of the metastatic lymph node ratio in patients with resectable carcinoma of ampulla of Vater. Medicine (Baltimore). 2015;94(42):e1859.
O’Connell JB, Maggard MA, Manunga J Jr, Tomlinson JS, Reber HA, Ko CY, et al. Survival after resection of ampullary carcinoma: a national population-based study. Ann Surg Oncol. 2008;15(7):1820–7.
Tol JA, Brosens LA, van Dieren S, van Gulik TM, Busch OR, Besselink MG, et al. Impact of lymph node ratio on survival in patients with pancreatic and periampullary cancer. Br J Surg. 2015;102(3):237–45.
Henson DE, Schwartz AM, Nsouli H, Albores-Saavedra J. Carcinomas of the pancreas, gallbladder, extrahepatic bile ducts, and ampulla of vater share a field for carcinogenesis: a population-based study. Arch Pathol Lab Med. 2009;133(1):67–71.
Berberat PO, Kunzli BM, Gulbinas A, Ramanauskas T, Kleeff J, Muller MW, et al. An audit of outcomes of a series of periampullary carcinomas. Eur J Surg Oncol. 2009;35(2):187–91.
Ahn DH, Bekaii-Saab T. Ampullary cancer: an overview. Am Soc Clin Oncol Educ Book. 2014:112–5.
Narang AK, Miller RC, Hsu CC, Bhatia S, Pawlik TM, Laheru D, et al. Evaluation of adjuvant chemoradiation therapy for ampullary adenocarcinoma: the Johns Hopkins Hospital-Mayo Clinic collaborative study. Radiat Oncol. 2011;6:126.
Howe JR, Klimstra DS, Moccia RD, Conlon KC, Brennan MF. Factors predictive of survival in ampullary carcinoma. Ann Surg. 1998;228(1):87–94.
Roder JD, Schneider PM, Stein HJ, Siewert JR. Number of lymph node metastases is significantly associated with survival in patients with radically resected carcinoma of the ampulla of Vater. Br J Surg. 1995;82(12):1693–6.
Su CH, Shyr YM, Lui WY, P'eng FK. Factors affecting morbidity, mortality and survival after pancreaticoduodenectomy for carcinoma of the ampulla of Vater. Hepatogastroenterology. 1999;46(27):1973–9.
Duffy JP, Hines OJ, Liu JH, Ko CY, Cortina G, Isacoff WH, et al. Improved survival for adenocarcinoma of the ampulla of Vater: fifty-five consecutive resections. Arch Surg. 2003;138(9):941–8 discussion 8-50.
Carter JT, Grenert JP, Rubenstein L, Stewart L, Way LW. Tumors of the ampulla of vater: histopathologic classification and predictors of survival. J Am Coll Surg. 2008;207(2):210–8.
Chang DK, Jamieson NB, Johns AL, Scarlett CJ, Pajic M, Chou A, et al. Histomolecular phenotypes and outcome in adenocarcinoma of the ampulla of vater. J Clin Oncol. 2013;31(10):1348–56.
Farid SG, Falk GA, Joyce D, Chalikonda S, Walsh RM, Smith AM, et al. Prognostic value of the lymph node ratio after resection of periampullary carcinomas. HPB (Oxford). 2014;16(6):582–91.
Falconi M, Crippa S, Domínguez I, Barugola G, Capelli P, Marcucci S, et al. Prognostic relevance of lymph node ratio and number of resected nodes after curative resection of ampulla of Vater carcinoma. Ann Surg Oncol. 2008;15(11):3178–86.
Kwon J, Kim K, Chie EK, Kim BH, Jang JY, Kim SW, et al. Prognostic relevance of lymph node status for patients with ampullary adenocarcinoma after radical resection followed by adjuvant treatment. Eur J Surg Oncol. 2017;43(9):1690–6.
Albagali RO, de Carvalho GSS, Mali J Jr, Eulalio JMR, de Melo ELR. Estudo da linfadenectomia radical comparada à linfadenectomia standard no tratamento cirúrgico do adenocarcinoma da papila de Vater. Rev Col Bras Cir. 2010;37(6):420–5.
Chen SC, Shyr YM, Chou SC, Wang SE. The role of lymph nodes in predicting the prognosis of ampullary carcinoma after curative resection. World J Surg Oncol. 2015;13:224.
Sierzega M, Nowak K, Kulig J, Matyja A, Nowak W, Popiela T. Lymph node involvement in ampullary cancer: the importance of the number, ratio, and location of metastatic nodes. J Surg Oncol. 2009;100(1):19–24.
Sakata J, Shirai Y, Wakai T, Ajioka Y, Akazawa K, Hatakeyama K. Assessment of the nodal status in ampullary carcinoma: the number of positive lymph nodes versus the lymph node ratio. World J Surg. 2011;35(9):2118–24.
Choi SB, Kim WB, Song TJ, Suh SO, Kim YC, Choi SY. Surgical outcomes and prognostic factors for ampulla of vater cancer. Scand J Surg. 2011;100(2):92–8.
Kim RD, Kundhal PS, McGilvray ID, Cattral MS, Taylor B, Langer B, et al. Predictors of failure after pancreaticoduodenectomy for ampullary carcinoma. J Am Coll Surg. 2006;202(1):112–9.
Barauskas G, Gulbinas A, Pranys D, Dambrauskas Z, Pundzius J. Tumor-related factors and patient’s age influence survival after resection for ampullary adenocarcinoma. J Hepato-Biliary-Pancreat Surg. 2008;15(4):423–8.
Palta M, Patel P, Broadwater G, Willett C, Pepek J, Tyler D, et al. Carcinoma of the ampulla of Vater: patterns of failure following resection and benefit of chemoradiotherapy. Ann Surg Oncol. 2012;19(5):1535–40.
Kim K, Chie EK, Jang JY, Kim SW, Oh DY, Im SA, et al. Role of adjuvant chemoradiotherapy for ampulla of Vater cancer. Int J Radiat Oncol Biol Phys. 2009;75(2):436–41.
Robert PE, Leux C, Ouaissi M, Miguet M, Paye F, Merdrignac A, et al. Predictors of long-term survival following resection for ampullary carcinoma: a large retrospective French multicentric study. Pancreas. 2014;43(5):692–7.
de Paiva Haddad LB, Patzina RA, Penteado S, Montagnini AL, da Cunha JE, Machado MC, et al. Lymph node involvement and not the histophatologic subtype is correlated with outcome after resection of adenocarcinoma of the ampulla of vater. J Gastrointest Surg. 2010;14(4):719–28.
Westgaard A, Tafjord S, Farstad IN, Cvancarova M, Eide TJ, Mathisen O, et al. Pancreatobiliary versus intestinal histologic type of differentiation is an independent prognostic factor in resected periampullary adenocarcinoma. BMC Cancer. 2008;8:170.
Roh YH, Kim YH, Lee HW, Kim SJ, Roh MS, Jeong JS, et al. The clinicopathologic and immunohistochemical characteristics of ampulla of Vater carcinoma: the intestinal type is associated with a better prognosis. Hepatogastroenterology. 2007;54(78):1641–4.
Ang DC, Shia J, Tang LH, Katabi N, Klimstra DS. The utility of immunohistochemistry in subtyping adenocarcinoma of the ampulla of vater. Am J Surg Pathol. 2014;38(10):1371–9.
Kwon MJ, Kim JW, Jung JP, Cho JW, Nam ES, Cho SJ, et al. Low incidence of KRAS, BRAF, and PIK3CA mutations in adenocarcinomas of the ampulla of Vater and their prognostic value. Hum Pathol. 2016;50:90–100.
Jun SY, Sung YN, Lee JH, Park KM, Lee YJ, Hong SM. Validation of the Eighth American Joint Committee on Cancer Staging System for distal bile duct carcinoma. Cancer Res Treat. 2019;51(1):98–111.
Huang XT, Huang CS, Chen W, Cai JP, Gan TT, Zhao Y, et al. Development and validation of a nomogram for predicting overall survival of node-negative ampullary carcinoma. J Surg Oncol. 2020;121(3):518–23.
Li HB, Zhao FQ, Zhou J. Prognostic nomogram for disease-specific survival in patients with non-metastatic ampullary carcinoma after surgery. Ann Surg Oncol. 2019;26(4):1079–85.
Acknowledgments
We thank Stela Maris de Jezus Castro and Sidia Jacques for statistical analysis support.
Funding
This work was supported by CNPq/Rede Genoprot (Grant #559814/2009-7) and Research and Events Incentive Fund (Fundo de Incentivo à Pesquisa e Eventos – FIPE, GPPG Grant #09-287) of Hospital de Clínicas de Porto Alegre (HCPA).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Vilhordo, D.W., Gregório, C., Valentini, D.F. et al. Prognostic Factors of Long-term Survival Following Radical Resection for Ampullary Carcinoma. J Gastrointest Canc 52, 872–881 (2021). https://doi.org/10.1007/s12029-020-00479-9
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12029-020-00479-9