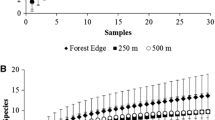
Abstract
Urbanisation is fast-growing worldwide characterised by the conversion of natural vegetation ecosystems into densely paved areas with a high concentration of human constructions and few remnants of natural habitats. This phenomenon may threaten wildlife, especially high trophic level organisms such as predators and parasitoids, which are known to be more sensitive to habitat fragmentation. We investigated the influence of urbanisation on the community of trap-nesting bees and wasps as well as on their parasites in an urban area. Trap-nests were installed in 11 areas within the perimeter of the city of Ribeirão Preto, Brazil. Fourteen land cover categories were distinguished and their percentages calculated for each area from satellite images. The community sampled consisted of 20 wasp and 12 bee species, as well as 25 natural enemy species that attacked 9.75% of the nests. The highest diversity of bees and wasps was observed in areas with higher percentages of natural vegetation, i.e., forests, wastelands, and neighbourhoods with extensive green areas. Rates of parasitism, measured by the abundance and richness of parasites, was positively correlated with the proportion of green areas in the landscape. Even though predatory wasps constitute a higher trophic level than bees they were not more negatively impacted by urbanisation. Our results demonstrate that natural habitats and extensive green areas can host diverse communities of cavity-nesting bees, wasps, and their parasites within a city. The conservation of green areas in urbanised landscapes should be considered as essential to maintain the populations of these important insects.


Similar content being viewed by others
References
Alaruikka D, Kotze DJ, Matveinen K, Niemelä J (2002) Carabid beetle and spider assemblages along a forested urban–rural gradient in southern Finland. J Insect Conserv 6(4):195–206
Albrecht M, Duelli P, Schmid B, Müller CB (2007) Interaction diversity within quantified insect food webs in restored and adjacent intensively managed meadows. J Anim Ecol 76(5):1015–1025
Araújo GJ, Fagundes R, Antonini Y (2018) Trap-nesting Hymenoptera and their network with parasites in recovered riparian forests Brazil. Neotrop Entomol 47(1):26–36
Araújo GJ, Storck-Tonon D, Izzo TJ (2019) Is planting trees enough? The effect of different types of reforestation on the offspring of Trypoxylon (Trypargilum) lactitarse (Hymenoptera: Crabronidae) in the Southern Amazon. Neotrop Entomol 48(4):572–582
Astete S, Marinho-Filho J, Kajin M, Penido G, Zimbres B, Sollmann R, Jácomo ATA, Tôrres NM, Silveira T (2017) Forced neighbours: coexistence between jaguars and pumas in a harsh environment. J Arid Environ 146:27–34
Banaszak-Cibicka W, Żmihorski M (2012) Wild bees along an urban gradient: winners and losers. J Insect Conserv 16(3):331–343
Bohart RM, Menke AS (1976) Sphecid wasps of the world: a generic revision. University of California Press, Berkeley
Bolker BM, R Development Core Team R (2012) Tools for general maximum likelihood estimation. R package version 1.0.5.2.
Boscolo D, Tokumoto PM, Ferreira PA, Ribeiro JW, Santos JS (2017) Positive responses of flower visiting bees to landscape heterogeneity depend on functional connectivity levels. Perspect Ecol Conserv 15(1):18–24
Burdine JD, McCluney KE (2019) Interactive effects of urbanization and local habitat characteristics influence bee communities and flower visitation rates. Oecologia. https://doi.org/10.1007/s00442-019-04416-x
Burnham KP, Anderson DR (2002) Model selection and multimodel inference: a practical information-theoretic approach, 2nd edition. Ecol Modell. https://doi.org/10.1016/J.ECOLMODEL.2003.11.004
Camillo E, Garófalo CA, Serrano JC, Muccillo G (1995) Diversidade e abundância sazonal de abelhas e vespas solitárias em ninhos armadilhas (Hymenoptera: Apocrita, Aculeata). Rev Bras Entomol 39(2):459–470
Cândido MEMB, Morato EF, Storck-Tonon D, Miranda PN, Vieira LJS (2018) Effects of fragments and landscape characteristics on the orchid bee richness (Apidae: Euglossini) in an urban matrix, southwestern Amazonia. J Insect Conserv 22(3–4):475–486
Cane JH, Minckley RL, Kervin LJ, Roulston TH, Williams NM (2006) Complex responses within a desert bee guild (Hymenoptera: Apiformes) to urban habitat fragmentation. Ecol Appl 16(2):632–644
Caryl FM, Lumsden LF, van der Ree R, Wintle BA (2016) Functional responses of insectivorous bats to increasing housing density support 'land-sparing' rather than 'land-sharing' urban growth strategies. J Appl Ecol 53(1):191–201
Chace JF, Walsh JJ (2006) Urban effects on native avifauna: a review. Landsc Urban Plan 74(1):46–69
Choate BA, Hickman PL, Moretti EA (2018) Wild bee species abundance and richness across an urban–rural gradient. J Insect Conserv 22(3–4):391–403
Clergeau P, Croci S, Jokimäki J, Kaisanlahti-Jokimäki ML, Dinetti M (2006) Avifauna homogenisation by urbanisation: analysis at different European latitudes. Biol Conserv 127(3):336–344
Collado MÁ, Sol D, Bartomeus I (2019) Bees use anthropogenic habitats despite strong natural habitat preferences. Divers Distrib 25(6):924–935
Conrad KF, Warren MS, Fox R, Parsons MS, Woiwod IP (2006) Rapid declines of common, widespread British moths provide evidence of an insect biodiversity crisis. Biol Conserv 132(3):279–291
Czech B, Krausman PR, Devers PK (2000) Economic associations among causes of species endangerment in the United States. Bioscience 50(7):593–601
Devictor V, Julliard R, Couvet D, Lee A, Jiguet F (2007) Functional homogenization effect of urbanization on bird communities. Conserv Biol 21(3):741–751
Dobson A, Lodge D, Alder J, Cumming GS, Keymer J, McGlade J, Mooney H, Rusak JA, Sala O, Wolters V, Wall D, Winfree R, Xenopoulos MA (2006) Habitat loss, trophic collapse, and the decline of ecosystem services. Ecology 87(8):1915–1924
Fischer LK, Eichfeld J, Kowarik I, Buchholz S (2016) Disentangling urban habitat and matrix effects on wild bee species. PeerJ 4:e2729
Fitch G, Glaum P, Simão MC, Vaidya C, Matthijs J, Iuliano B, Perfecto I (2019) Changes in adult sex ratio in wild bee communities are linked to urbanization. Sci Rep 9:3767
Flores LMA, Zanette LRS, Araujo FS (2018) Effects of habitat simplification on assemblages of cavity nesting bees and wasps in a semiarid Neotropical conservation area. Biodivers Conserv 27(2):311–328
Fortel L, Henry M, Guibaud L, Guirao AL, Kuhlmann M, Mouret H, Rollin O, Vaissière BE (2014) Decreasing abundance, increasing diversity and changing structure of the wild bee community (Hymenoptera: Anthophila) along an urbanization gradient. PLoS ONE 9(8):e104679
Freiria GA, Rocha-Filho LC, Del Lama MA, Garófalo CA (2017) Surrounded by foes: Parasitism in Euglossa spp. (Apidae, Euglossini) caused by six species of natural enemies. J Apicult Res 56(3):255–261
Germaine SS, Wakeling BF (2001) Lizard species distributors and habitat occupation along an urban gradient in Tucson, Arizona, USA. Biol Conserv 97(2):229–237
Geslin B, Le Féon V, Folschweiller M, Flacher F, Carmignac D, Motard E, Perret S, Dajoz I (2016) The proportion of impervious surfaces at the landscape scale structures wild bee assemblages in a densely populated region. Ecol Evol 6(18):6599–6615
Gibb D, Hochuli DF (2002) Habitat fragmentation in an urban environment: large and small fragments support different arthropod assemblages. Biol Conserv 106(1):91–100
Google Inc. (2018) Google Maps. 2018. https://maps.google.com. Accessed 19 May 2018
Guenat S, Kunin WE, Dougill AJ, Dallimer M (2019) Effects of urbanisation and management practices on pollinators in tropical Africa. J Appl Ecol 56(1):214–224
Guzzo P, Carneiro RMA, Oliveira-Júnior H (2006) Cadastro municipal de espaços livres urbanos de Ribeirão Preto (SP): acesso público, índices e base para novos instrumentos e mecanismos de gestão. Revista da Sociedade Brasileira de Arborização Urbana 1(1):19–30
Hammer Ø, Harper DAT, Ryan PD (2001) PAST: paleontological statistics software package for education and data analysis. Palaeontol Electron 4(1):1–9
Hass AL, Brachmann L, Batáry P, Clough Y, Behling H, Tscharntke T (2019) Maize-dominated landscapes reduce bumblebee colony growth through pollen diversity loss. J Appl Ecol 56(2):294–304
Hernandez JL, Frankie GW, Thorp RW (2009) Ecology of urban bees: a review of current knowledge and directions for future study. Cities Environ 2(1):360–376
Iantas J, Woitowicz FCG, Buschini MLT (2017) Habitat modification and alpha-beta diversity in trap nesting bees and wasps (Hymenoptera: Aculeata) in southern Brazil. Trop Zool 30(2):83–96
IBGE - Instituto Brasileiro de Geografia e Estatística (2019) Cidades, IBGE, Rio de Janeiro. https://cidades.ibge.gov.br/v4/brasil/sp/ribeirao-preto/panorama. Accessed 11 Dec 2019
Kotchetkoff-Henriques O, Joly CA, Bernacci LC (2005) Soil and floristic composition of native vegetation remnants relationship in the municipality of Ribeirão Preto SP. Rev Bras Bot 28(3):541–562
Krauss J, Bommarco R, Guardiola M, Heikkinen RK, Helm A, Kuussaari M, Lindborg R, Öckinger E, Pärtel M, Pino J, Pöyry J, Raatikainen KM, Sang A, Stefanescu C, Teder T, Zobel M, Steffan-Dewenter I (2010) Habitat fragmentation causes immediate and time-delayed biodiversity loss at different trophic levels. Ecol Lett 13(5):597–605
Krewenka KM, Holzschuh A, Tscharntke T, Dormann CF (2011) Landscape elements as potential barriers and corridors for bees, wasps and parasitoids. Biol Conserv 144(6):1816–1825
Krombein KV (1967) Trap-nesting wasps and bees: life histories, nests, and associates. Smithsonian Institutional Press, Washington
Kruess A, Tscharntke T (2000) Species richness and parasitism in a fragmented landscape: experiments and field studies with insects on Vicia sepium. Oecologia 122(1):129–137
La Salle J, Gauld ID (1993) Hymenoptera and biodiversity. CAB International, Wallingford
Lassau SA, Hochuli DF (2007) Associations between wasp communities and forest structure: Do strong local patterns hold across landscapes? Aust Ecol 32(6):656–662
Lerman SB, Milam J (2016) Bee fauna and floral abundance within lawn-dominated suburban yards in Springfield, MA. Ann Entomol Soc Am 109(5):713–723
MacIvor JS (2016) Building height matters: nesting activity of bees and wasps on vegetated roofs. Isr J Ecol Evol 62(1–2):88–96
MacIvor JS (2017) Cavity-nest boxes for solitary bees: A century of design and research. Apidologie 48(3):311–327
MacIvor JS, Packer L (2015) ‘Bee hotels’ as tools for native pollinator conservation: a premature verdict? PLoS ONE 10(3):e0122126
MacIvor JS, Packer L (2016) The bees among us: modelling occupancy of solitary bees. PLoS ONE 11(12):e0164764
Magurran AE (2004) Measuring biological diversity. Blackwell, Oxford
Mallinger RE, Gibbs J, Gratton C (2016) Diverse landscapes have a higher abundance and species richness of spring wild bees by providing complementary floral resources over bees' foraging periods. Landsc Ecol 31(7):1523–1535
Matos MCB, Silva SS, Teodoro AV (2016) Seasonal population abundance of the assembly of solitary wasps and bees (Hymenoptera) according to land-use in Maranhão state, Brazil. Rev Bras Entomol 60(2):171–176
Matteson KC, Ascher JS, Langellotto GA (2008) Bee richness and abundance in New York City urban gardens. Ann Entomol Soc Am 101(1):140–150
McIntyre NE (2000) Ecology of urban arthropods: a review and a call to action. Ann Entomol Soc Am 93:825–835
McGarigal K, Cushman SA, Ene E (2012) FRAGSTATS v4: spatial pattern analysis program for categorical and continuous maps. In: Computer software program produced by the authors at the University of Massachusetts, Amherst
McIntyre NE, Rango J, Fagan WF, Faeth SH (2001) Ground arthropod community structure in a heterogeneous urban environment. Landsc Urban Plan 52(4):257–274
McKinney ML (2006) Urbanization as a major cause of biotic homogenization. Biol Conserv 127(3):247–260
McKinney ML (2008) Effects of urbanization on species richness: a review of plants and animals. Urban Ecosyst 11(2):161–176
Michener CD (2007) The bees of the World, 2nd edn. Johns Hopkins University Press, Baltimore
Minckley RL, Danforth BN (2019) Sources and frequency of brood loss in solitary bees. Apidologie 50(4):515–525
Molumby A (1997) Why make daughters larger? Maternal sex-allocation and sex-dependent selection for body size in a mass-provisioning wasp, Trypoxylon politum. Behav Ecol 8(3):279–287
Nelson AE, Forbes AA (2014) Urban land use decouples plant-herbivore-parasitoid interactions at multiple spatial scales. PLoS ONE 9(7):e102127
Nether MC, Dudek J, Buschini MLT (2019) Trophic interaction and diversity of cavity-nesting bees and wasps (Hymenoptera: Aculeata) in Atlantic forest fragments and in adjacent matrices. Apidologie 50(1):104–115
Normandin É, Vereecken NJ, Buddle CM, Fournier V (2017) Taxonomic and functional trait diversity of wild bees in different urban settings. PeerJ 5:e3051
Ollerton J, Winfree R, Tarrant S (2011) How many flowering plants are pollinated by animals? Oikos 120(3):321–326
Ostfeld RS, LoGiudice K (2003) Community disassembly, biodiversity loss, and the erosion of an ecosystem service. Ecology 84(6):1421–1427
Pereira-Peixoto MH, Pufal G, Staab M, Martins CF, Klein AM (2016) Diversity and specificity of host-natural enemy interactions in an urban-rural interface. Ecol Entomol 41(3):241–252
Perillo LN, Neves FS, Antonini Y, Martins RP (2017) Compositional changes in bee and wasp communities along Neotropical mountain altitudinal gradient. PLoS ONE 12(7):e0182054
QGIS Development Team (2017) QGIS Geographic Information System. Open Source Geospatial Foundation. https://www.qgis.org. Accessed 10 May 2019
Rocha EA, Fellowes MDE (2018) Does urbanization explain differences in interactions between an insect herbivore and its natural enemies and mutualists? Urban Ecosyst 21(3):405–417
Rocha-Filho LC, Rabelo LS, Augusto SC, Garófalo CA (2017) Cavity-nesting bees and wasps (Hymenoptera: Aculeata) in a semi-deciduous Atlantic forest fragment immersed in a matrix of agricultural land. J Insect Conserv 21(4):727–736
Rochat E, Manel S, Deschamps-Cottin M, Widmer I, Joost S (2017) Persistence of butterfly populations in fragmented habitats along urban density gradients: motility helps. Heredity 119(5):328–338
RStudio Team (2015) RStudio: Integrated Development for R. RStudio, Inc., Boston. https://www.rstudio.com/. Accessed 10 May 2019
Sala OE, Stuart-Chapin F III, Armesto JJ, Berlow E, Bloomfield J, Dirzo R, Huber-Sanwald E, Huenneke LF, Jackson RB, Kinzig A, Leemans R, Lodge DM, Mooney HA, Oesterheld M, Poff NL, Sykes MT, Walker BH, Walker M, Wall DH (2000) Global biodiversity scenarios for the year 2100. Science 287(5459):1770–1774
Santoni MM, Brescovit AD, Del Lama MA (2009) Ocupação diferencial do habitat por vespas do gênero Trypoxylon (Trypargilum) Latreille (Hymenoptera, Crabronidae). Rev Bras Entomol 53(1):107–114
Seto KC, Fragkias M, Güneralp B, Reilly MK (2011) A meta-analysis of global urban land expansion. PLoS ONE 6(8):e23777
Sheffield CS, Pindar A, Packer L, Kevan PG (2013) The potential of cleptoparasitic bees as indicator taxa for assessing bee communities. Apidologie 44(5):501–510
Shochat E, Stefanov WL, Whitehouse ME, Faeth SH (2004) Urbanization and spider diversity: influences of human modification of habitat structure and productivity. Ecol Appl 14(1):268–280
Silva FO, Viana BF, Boscolo D, Santos RL (2019) Foraging activity of Xylocopa cearensis (Ducke) in sand dune landscape. Sociobiology 66(1):190–193
Silva LG, Ribeiro MC, Hasui E, Costa CA, Cunha RGT (2015) Patch size, functional isolation, visibility and matrix permeability influences Neotropical primate occurrence within highly fragmented landscapes. PLoS ONE 10(2):e0114025
Smith RM, Warren PH, Thompson K, Gaston KJ (2006) Urban domestic gardens (VI): environmental correlates of invertebrate species richness. Biodivers Conserv 15(8):2415–2438
Soga M, Yamaura Y, Koike S, Gaston KJ (2014) Land sharing vs. land sparing: Does the compact city reconcile urban development and biodiversity conservation? J Appl Ecol 51(5):1378–1386
Stangler ES, Hanson PE, Steffan-Dewenter I (2015) Interactive effects of habitat fragmentation and microclimate on trap-nesting Hymenoptera and their trophic interactions in small secondary rainforest remnants. Biodivers Conserv 24(3):563–577
Steffan-Dewenter I, Münzenberg U, Bürger C, Thies C, Tscharntke T (2002) Scale-dependent effects of landscape context on three pollinator guilds. Ecology 83(5):1421–1432
Stewart AB, Sritongchuay T, Teartisup P, Kaewsomboon S, Bumrungsri S (2018) Habitat and landscape factors influence pollinators in a tropical megacity, Bangkok. Thailand PeerJ 6:e5335
Threlfall CG, Walker K, Williams NSG, Hahs AK, Mata L, Stork N, Livesley SJ (2015) The conservation value of urban green space habitats for Australian native bee communities. Biol Conserv 187:240–248
Tscharntke T, Gathmann A, Dewenter IS (1998) Bioindication using trap-nesting bees and wasps and their natural enemies: community structure and interactions. J Appl Ecol 35(5):708–719
Villaseñor NR, Driscoll DA, Escobar MAH, Gibbons P, Lindenmayer DB (2014) Urbanization impacts on mammals across urban-forest edges and a predictive model of edge effects. PLoS ONE 9(5):e97036
Vitousek PM, Mooney HA, Lubchenco J, Melillo J (1997) Human domination of Earth's ecosystem. Science 277(5325):494–499
Zabel J, Tscharntke T (1998) Does fragmentation of Urtica habitats affect phytophagous and predatory insects differentially? Oecologia 116(3):419–425
Zanette LRS, Martins RP, Ribeiro SP (2005) Effects of urbanization on Neotropical wasp and bee assemblages in a Brazilian metropolis. Landsc Urban Plan 71(2–4):105–121
Zar JH (1996) Biostatistical analysis. Prentice-Hall, Upper Saddle River
Zurbuchen A, Landert L, Klaiber J, Müller A, Hein S, Dorn S (2010) Maximum foraging ranges in solitary bees: only few individuals have the capability to cover long foraging distances. Biol Conserv 143(3):669–676
Acknowledgements
The authors are grateful to the following taxonomists each for identifying some of the sampled species: Cecília Waichert (Pompilidae), Alexandre Aguiar (Ichneumonidae), Marcelo Tavares (Chalcididae), Daercio Lucena (Chrysididae), Daniele Parizotto (Dicranthidium), Jan Batelka (Ripiphoridae), Marcos Kogan (Xenidae), Barry O'Connor (Pyemotidae), Thiago Izzo (Formicidae), Rogério Lopes and Bolívar Garcete-Barrett (some Vespidae). We thank 'Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—CAPES' for providing a scholarship to the first author, Laurence Packer for his careful reading and constructive comments that improved the manuscript, Heraldo Vasconcelos for his valuable assistance with some statistical analysis and all the people who authorised fieldwork in the sampling areas.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Jens Wolfgang Dauber.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article belongs to the Topical Collection: Urban biodiversity.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary file4 (TIF 2294 kb)
Fig. S1. A - Metallic support containing the trap nests. Two wooden plates with 110 black cardboard tubes are located right below the plastic tile whilst 120 bamboo canes are placed within three PVC tubes below. B - MDF box with external measurements of 8 cm x 8 cm x 5 cm and a side hole of 1.2 cm diameter. Two boxes were placed between the two wooden plates (red arrow in A). C - Two Ambay pumpwood petioles attached vertically to the oil chamber of the metallic support (blue arrow in A).
Supplementary file5 (TIF 29178 kb)
Fig. S2. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the Landscape resistance index (LRI) as the predictor variable for bee variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file6 (TIF 29178 kb)
Fig. S3. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the functional connectivity of green areas (GAC) as the predictor variable for bee variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file7 (TIF 29178 kb)
Fig. S4. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the proportion of mixed areas as the predictor variable for bee variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file8 (TIF 29178 kb)
Fig. S5. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the proportion of green areas as the predictor variable for bee variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file9 (TIF 29178 kb)
Fig. S6. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the Landscape resistance index (LRI) as the predictor variable for wasp variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file10 (TIF 29178 kb)
Fig. S7. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the proportion of mixed areas as the predictor variable for wasp variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file11 (TIF 29178 kb)
Fig. S8. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the proportion of green areas as the predictor variable for wasp, natural enemy and whole community variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file12 (TIF 29178 kb)
Fig. S9. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the proportion of mixed areas as the predictor variable for natural enemy variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file13 (TIF 29178 kb)
Fig. S10. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the Landscape resistance index (LRI) as the predictor variable for natural enemy and whole community variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file14 (TIF 29178 kb)
Fig. S11. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the functional connectivity of green areas (GAC) as the predictor variable for wasp, natural enemy, caterpillar- and spider-hunting wasp variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file15 (TIF 13613 kb)
Fig. S12. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the proportion of mixed areas as the predictor variable for whole community variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file16 (TIF 29178 kb)
Fig. S13. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the Landscape resistance index (LRI) as the predictor variable for caterpillar-hunting wasp variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file17 (TIF 29178 kb)
Fig. S14. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the proportion of green areas as the predictor variable for caterpillar- and spider-hunting wasp variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file18 (TIF 29178 kb)
Fig. S15. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the proportion of mixed areas as the predictor variable for caterpillar-hunting wasp variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file19 (TIF 29178 kb)
Fig. S16. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the Landscape resistance index (LRI) as the predictor variable for spider-hunting wasp variables. Dots indicate the sampled areas used in the GLMs.
Supplementary file20 (TIF 13613 kb)
Fig. S17. Best selected Generalised Linear Models (GLMs) according to the second order Akaike's Information Criterion (AICc) with the proportion of mixed areas as the predictor variable for spider-hunting wasp variables. Dots indicate the sampled areas used in the GLMs.
Rights and permissions
About this article
Cite this article
da Rocha-Filho, L.C., Montagnana, P.C., Boscolo, D. et al. Green patches among a grey patchwork: the importance of preserving natural habitats to harbour cavity-nesting bees and wasps (Hymenoptera) and their natural enemies in urban areas. Biodivers Conserv 29, 2487–2514 (2020). https://doi.org/10.1007/s10531-020-01985-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10531-020-01985-9