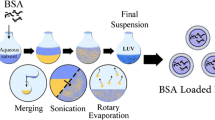
Abstract
We first demonstrate a simple and rapid fabrication protocol of bovine serum albumin (BSA) nanoparticles (NPs), as potential drug carriers using a microchannel flow technique with the successful encapsulation of a water-soluble kynurenic acid (KYNA) having neuroactive property has also been performed. By comparison, the preparation of a hydrophobic α-Tocopherol (TP)-loaded polylactide-co-glycolide 50:50 (PLGA50)-based NPs was also carried out under flow conditions. We highlight several benefits of the flow technique over the commonly known self-assembly and nanoprecipitation processes. The average particle diameter, the size distribution, the encapsulation efficiency (EE%) and the drug release kinetics of these different core-shell type NPs prepared by the flow as well as the above-mentioned classic methods were compared. The decisive role of the flow rate (FR), the relative flow rate (RFR) of the components in the particle size of both BSA- and PLGA50-based NPs have also been verified. By utilization of the optimal flow conditions, the average size can be decreased with ca. 15–20% and lower polydispersity index (PDI) can be also achieved. It was confirmed that the systematic change of the RFR values resulting in the controlled size of the drug-loaded BSA NPs between 120 and 140 nm, while d = 149 nm was obtained for self-assembled NPs. However, for BSA-based NPs quite similar EE% was obtained for both methods (ca. 11–12%), but for PLGA50/TP NPs the application of flow device increased the EE% from 67.0% to 71.5%.





Similar content being viewed by others
References
Nicolas J, Mura S, Brambilla D, MacKiewicz N, Couvreur P (2013) Design, functionalization strategies and biomedical applications of targeted biodegradable/biocompatible polymer-based nanocarriers for drug delivery. Chem Soc Rev 42:1147–1235
Merodio M, Arnedo A, Renedo MJ, Irache JM (2001) Ganciclovir-loaded albumin nanoparticles: characterization and in vitro release properties. Eur J Pharm Sci 12:251–259
De Paoli Lacerda SH, Ingber B, Rosenzweig N (2005) Structure-release rate correlation in collagen gels containing fluorescent drug analog. Biomaterials 26:7164–7172
Elzoghby AO, Samy WM, Elgindy NA (2012) Albumin-based nanoparticles as potential controlled release drug delivery systems. J Control Release 157:168–182
Donsì F, Senatore B, Huang Q, Ferrari G (2010) Development of novel pea protein-based nanoemulsions for delivery of nutraceuticals. J Agric Food Chem 58:10653–10660
Mehnert W, Mäder K (2001) Solid lipid nanoparticles: production, characterization and applications. Adv Drug Deliv Rev 47:165–196
Chunfu Z, Jinquan C, Duanzhi Y, Yongxian W, Yanlin F, Jiajü T (2004) Preparation and radiolabeling of human serum albumin (HSA)-coated magnetite nanoparticles for magnetically targeted therapy. Appl Radiat Isot 61:1255–1259
Pinto Reis C, Neufeld RJ, Ribeiro AJ, Veiga F (2006) Nanoencapsulation I. Methods for preparation of drug-loaded polymeric nanoparticles. Nanomed Nanotechnol Biol Med 2:8–21
Yu S, Yao P, Jiang M, Zhang G (2006) Nanogels prepared by self-assembly of oppositely charged globular proteins. Biopolymers 83:148–158
Lee SH, Heng D, Ng WK, Chan HK, Tan RBH (2011) Nano spray drying: a novel method for preparing protein nanoparticles for protein therapy. Int J Pharm 403:192–200
Varca GHC, Ferraz CC, Lopes PS, Beatriz Mathor M, Grasselli M, Lugão AB (2013) Radio-synthesized protein-based nanoparticles for biomedical purposes. Radiat Phys Chem 94:181–185
Kim TH, Jiang HH, Youn YS, Park CW, Tak KK, Lee S, Kim H, Jon S, Chen X, Lee KC (2011) Preparation and characterization of water-soluble albumin-bound curcumin nanoparticles with improved antitumor activity. Int J Pharm 403:285–291
Ferrado JB, Perez AA, Visentini FF, Islan GA, Castro GR, Santiago LG (2019) Formation and characterization of self-assembled bovine serum albumin nanoparticles as chrysin delivery systems. Colloids Surf B Biointerfaces 173:43–51
Fraylich MR, Liu R, Richardson SM, Baird P, Hoyland J, Freemont AJ, Alexander C, Shakesheff K, Cellesi F, Saunders BR (2010) Thermally-triggered gelation of PLGA dispersions: towards an injectable colloidal cell delivery system. J Colloid Interface Sci 344:61–69
Govender T, Stolnik S, Garnett MC, Illum L, Davis SS (1999) PLGA nanoparticles prepared by nanoprecipitation: drug loading and release studies of a water soluble drug. J Control Release 57:171–185
Varga N, Turcsányi Á, Hornok V, Csapó E (2019) Vitamin E-loaded PLA- and PLGA-based core-shell nanoparticles: synthesis, structure optimization and controlled drug release. Pharmaceutics 11:357
Varga N, Hornok V, Janovák L, Dékány I, Csapó E (2019) The effect of synthesis conditions and tunable hydrophilicity on the drug encapsulation capability of PLA and PLGA nanoparticles. Colloids Surf B Biointerfaces 176:212–218
Karnik R, Gu F, Basto P, Cannizzaro C, Dean L, Kyei-Manu W, Langer R, Farokhzad OC (2008) Microfluidic platform for controlled synthesis of polymeric nanoparticles. Nano Lett 8:2906–2912
Capretto L, Carugo D, Cheng W, Hill M, Zhang X (2011) Continuous-flow production of polymeric micelles in microreactors: experimental and computational analysis. J Colloid Interf Sci 357:243–251
Jankowski P, Kutaszewicz R, Ogończyk D, Garstecki P (2019) A microfluidic platform for screening and optimization of organic reactions in droplets. J Flow Chem 8:1–12
Wojnicki M, Tokarski T, Hessel V, Fitzner K, Luty-Błocho M (2019) Continuous, monodisperse silver nanoparticles synthesis using microdroplets as a reactor. J Flow Chem 9:1–7
Vécsei L, Szalárdy L, Fülöp F, Toldi J (2013) Kynurenines in the CNS: recent advances and new questions. Nat Rev Drug Discov 12:64–82
Veres G, Molnár M, Zádori D, Szentirmai M, Szalárdy L, Török R, Fazekas E, Ilisz I, Vécsei L, Klivényi P (2015) Central nervous system-specific alterations in the tryptophan metabolism in the 3-nitropropionic acid model of Huntington’s disease. Pharmacol Biochem Behav 132:115–124
Varga N, Csapó E, Majláth Z, Ilisz I, Krizbai IA, Wilhelm I, Knapp L, Toldi J, Vécsei L, Dékány I (2016) Targeting of the kynurenic acid across the blood-brain barrier by core-shell nanoparticles. Eur J Pharm Sci 86:67–74
Varga N, Dékány I, Knapp L, Krizbai I, Majláth Zs, Toldi J, Vécsei L (2020) A process for the delivery of drug molecules through the blood-brain barrier using core-shell nanocomposites, accepted Hungarian patent, P1500356
Costa P, Sousa Lobo JM (2001) Modeling and comparison of dissolution profiles. Eur J Pharm Sci13:123–133
Dash S, Murthy PN, Nath L, Chowdhury P (2010) Kinetic modeling on drug release from controlled drug delivery systems. Acta Pol Pharm 67:217–223
Ritger PL, Peppas NA (1987) A simple equation for description of solute release I. Fickian and non-fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs. J Control Release 5:23–36
Murugeshu A, Astete C, Leonardi C, Morgan T, Sabliov CM (2011) Chitosan/PLGA particles for controlled release of α-tocopherol in the GI tract via oral administration. Nanomedicine 6:1513–1528
Astete CE, Dolliver D, Whaley M, Khachatryan L, Sabliov CM (2011) Antioxidant poly(lactic-co-glycolic) acid nanoparticles made with α-Tocopherol–ascorbic acid surfactant. ACS Nano 5:9313–9325
Zigoneanu IG, Astete CE, Sabliov CM (2008) Nanoparticles with entrapped α-tocopherol: synthesis, characterization, and controlled release. Nanotechnology 19:105606
Veres P, Kéri M, Bányai I, Lázár I, Domingo C, Kalmár J (2017) Mechanism of drug release from silica-gelatin aerogel-relationship between matrix structure and release kinetics. Colloids Surf B Biointerfaces 152:229–237
Kéri M, Forgács A, Papp V, Bányai I, Veres P, Len A, Dudás Z, Fábián I, Kalmár J (2020) Gelatin content governs hydration induced structural changes in silica-gelatin hybrid aerogels – implications in drug delivery. Acta Biomater
Acknowledgements
The research was supported by the National Research, Development and Innovation Office-NKFIH through the project GINOP-2.3.2-15-2016-00034 and FK 131446. V. Hornok acknowledges the Premium Post Doctorate Research Program of the Hungarian Academy of Sciences for the financial support. This paper was supported by the János Bolyai Research Scholarship of the Hungarian Academy of Sciences (E. Csapó). The Ministry of Human Capacities, Hungary grant TUDFO/47138-1/2019-ITM is acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
- Controllable preparation of serum albumin nanocarriers containing kynurenic acid has been presented under flow conditions
- By optimized flow conditions, a reproducible and large-scale production of BSA-based core-shell nanoparticles can be obtained
- Utilization of flow device results in smaller particle sizes compared to self-assembly and nanoprecipitation methods
Electronic supplementary material
ESM 1
(DOCX 770 kb)
Rights and permissions
About this article
Cite this article
Kovács, A.N., Varga, N., Gombár, G. et al. Novel feasibilities for preparation of serum albumin-based core-shell nanoparticles in flow conditions. J Flow Chem 10, 497–505 (2020). https://doi.org/10.1007/s41981-020-00088-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s41981-020-00088-4