Abstract
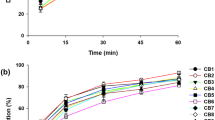
Triclabendazole belongs to the class II/IV of the Biopharmaceuticals Classification System, and its low aqueous solubility represents a major drawback during the development of effective dosage forms. Therefore, the goal of this study was to elucidate whether polymeric solid dispersions would represent a suitable approach to overcome such disadvantage. Due to the lack of information on triclabendazole release, four different dissolution media were evaluated to analyze drug dissolution rate. The polymeric solid dispersions were characterized by X-ray diffraction and Fourier transform infrared spectroscopy. The selected final formulations were further stored for 24 months, and their physical stability was evaluated by means of X-ray diffraction and drug dissolution assays. Drug solubility studies indicated that poloxamer 407 (P407) solubilized a higher amount of drug than polyethylene glycol 6000. Drug-to-carrier ratio, nature of the selected carriers, and the type of dissolution media were important factors for increasing dissolution. By infrared spectroscopy, there were no specific interactions between the drug and polymers. The physicochemical characterization of the systems showed a detectable evidence of drug amorphization by increasing the carrier ratio. Micromeritic studies indicated that raw triclabendazole, physical mixtures, and reference formulation showed poor flow properties, in contrast to the triclabendazole:P407 solid dispersion sample. Both the crystalline properties and dissolution rate of selected samples were very similar after 24 months at room temperature. Thus, considering physical stability and dissolution studies, the development of the solid dispersion is a very suitable methodology to improve triclabendazole dissolution and, potentially, its biopharmaceutical performance.










Similar content being viewed by others
References
Ashrafi K, Bargues MD, O’Neill S, Mas-Coma S. Fascioliasis: a worldwide parasitic disease of importance in travel medicine. Travel Med Infect Dis. 2014;12(6 Pt A):636–49. https://doi.org/10.1016/j.tmaid.2014.09.006.
Mas-Coma S, Bargues MD, Valero MA. Human fascioliasis infection sources, their diversity, incidence factors, analytical methods and prevention measures. Parasitology. 2018;145(13):1665–99. https://doi.org/10.1017/S0031182018000914.
20th WHO essential medicines list. 2017. https://www.who.int/medicines/news/2017/20th_essential_med-list/en/.
Keiser J, Engels D, Buscher G, Utzinger J. Triclabendazole for the treatment of fascioliasis and paragonimiasis. Expert Opin Investig Drugs. 2005;14(12):1513–26. https://doi.org/10.1517/13543784.14.12.1513.
Fairweather I. Triclabendazole progress report, 2005–2009: an advancement of learning? J Helminthol. 2009;83(2):139–50. https://doi.org/10.1017/S0022149X09321173.
Food and Drug Administration. https://www.accessdata.fda.gov/drugsatfda_docs/label/2019/208711s000lbl.pdf.
Cabada MM, Lopez M, Cruz M, Delgado JR, Hill V, White AC Jr. Treatment failure after multiple courses of triclabendazole among patients with fascioliasis in Cusco, Peru: a case series. PLoS Negl Trop Dis. 2016;10(1):e0004361. https://doi.org/10.1371/journal.pntd.0004361.
Mas-Coma S, Bargues MD, Valero MA. Diagnosis of human fascioliasis by stool and blood techniques: update for the present global scenario. Parasitology. 2014;141(14):1918–46. https://doi.org/10.1017/S0031182014000869.
Lindenberg M, Kopp S, Dressman JB. Classification of orally administered drugs on the World Health Organization model list of essential medicines according to the biopharmaceutics classification system. 2004;58(2):265–78. https://doi.org/10.1016/j.ejpb.2004.03.001.
Flores-Ramos M, Ibarra-Velarde F, Jung-Cook H, Hernández-Campos A, Vera-Montenegro Y, Castillo R. Novel triclabendazole prodrug: a highly water soluble alternative for the treatment of fasciolosis. Bioorg Med Chem Lett. 2017;27(3):616–9. https://doi.org/10.1016/j.bmcl.2016.12.004.
Luzardo-Álvarez A, Martínez-Mazagastos J, Santamarina-Fernández MT, Otero-Espinar FJ, Blanco-Méndez J. Oral pharmacological treatments for ichthyophthiriosis of rainbow trout (Oncorhynchus mykiss). Aquaculture. 2003;220(1-4):15–25. https://doi.org/10.1016/S0044-8486(02)00228-4.
Real D, Leonardi D, Williams RO III, Repka MA, Salomon CJ. Solving the delivery problems of triclabendazole using cyclodextrins. AAPS PharmSciTech. 2018;19(5):2311–21. https://doi.org/10.1208/s12249-018-1057-5.
Real D, Hoffmann S, Leonardi D, Salomon C, Goycoolea FM. Chitosan based nanodelivery systems applied to the development of novel triclabendazole formulations. PLoS One. 2018;13(12):e0207625. https://doi.org/10.1371/journal.pone.0207625.
Zhou Y, Du J, Wang L, Wang Y. Nanocrystals technology for improving bioavailability of poorly soluble drugs: a mini-review. J Nanosci Nanotechnol. 2017;17(1):18–28.
Seremeta KP, Arrúa EA, Okulik NB, Salomon CJ. Development and characterization of benznidazole nano- and microparticles: a new tool for pediatric treatment of Chagas disease? Colloids Surf B Biointerfaces. 2019;177:169–77. https://doi.org/10.1016/j.colsurfb.2019.01.039.
Bhatnagar P, Dhote V, Mahajan SC, Mishra PK, Mishra DK. Solid dispersion in pharmaceutical drug development: from basics to clinical applications. Curr Drug Deliv. 2014;11(2):155–71.
Chowdhury N, Vhora I, Patel K, Bagde A, Kutlehria S, Singh M. Development of hot melt extruded solid dispersion of tamoxifen citrate and resveratrol for synergistic effects on breast cancer cells. AAPS PharmSciTech. 2018;19(7):3287–97. https://doi.org/10.1208/s12249-018-1111-3.
Huang Y, Dai WG. Fundamental aspects of solid dispersion technology for poorly soluble drugs. Acta Pharm Sin B. 2014;4(1):18–25. https://doi.org/10.1016/j.apsb.2013.11.001.
Vo CL, Park C, Lee BJ. Current trends and future perspectives of solid dispersions containing poorly water-soluble drugs. Eur J Pharm Biopharm. 2013;85(3 Pt B):799–813. https://doi.org/10.1016/j.ejpb.2013.09.007.
Winslow CJ, Nichols BLB, Novo DC, Mosquera-Giraldo LI, Taylor LS, Edgar KJ, et al. Cellulose-based amorphous solid dispersions enhance rifapentine delivery characteristics in vitro. Carbohydr Polym. 2018;182:149–58. https://doi.org/10.1016/j.carbpol.2017.11.024.
Costa ED, Priotti J, Orlandi S, Leonardi D, Lamas MC, Nunes TG, et al. Unexpected solvent impact in the crystallinity of praziquantel/poly(vinylpyrrolidone) formulations. A solubility, DSC and solid state NMR study. Int J Pharm. 2016;511(2):983–93. https://doi.org/10.1016/j.ijpharm.2016.08.009.
Simonazzi A, Cid AG, Paredes AJ, Schofs L, Gonzo EE, Palma SD, et al. Development and in vitro evaluation of solid dispersions as strategy to improve albendazole biopharmaceutical behavior. Ther Deliv. 2018;9(9):623–38. https://doi.org/10.4155/tde-2018-0037.
Song CK, Yoon IS, Kim DD. Poloxamer-based solid dispersions for oral delivery of docetaxel: differential effects of F68 and P85 on oral docetaxel bioavailability. Int J Pharm. 2016;507(1-2):102–8. https://doi.org/10.1016/j.ijpharm.2016.05.002.
Leonardi D, Barrera MG, Lamas MC, Salomon CJ. Development of prednisone:polyethylene glycol 6000 fast-release tablets from solid dispersions: solid-state characterization, dissolution behavior, and formulation parameters. AAPS PharmSciTech. 2007;8(4):221. https://doi.org/10.1208/pt0804108.
Tajarobi F, Abrahmsén-Alami S, Larsson A. Dissolution rate enhancement of parabens in PEG solid dispersions and its influence on the release from hydrophilic matrix tablets. J Pharm Sci. 2011;100(1):275–83. https://doi.org/10.1002/jps.22248.
Fonseca-Berzal C, Palmeiro-Roldán R, Escario JA, Torrado S, Arán VJ, Torrado-Santiago S, et al. Novel solid dispersions of benznidazole: preparation, dissolution profile and biological evaluation as alternative antichagasic drug delivery system. Exp Parasitol. 2015;149:84–91. https://doi.org/10.1016/j.exppara.2015.01.002.
Jiménez de los Santos CJ, Pérez-Martínez JI, Gómez-Pantoja ME, Moyano JR. Enhancement of albendazole dissolution properties using solid dispersions with Gelucire 50/13 and PEG 15000. J Drug Deliv Sci Technol. 2017;42:261–72. https://doi.org/10.1016/j.jddst.2017.03.030.
García-Rodriguez JJ, de la Torre-Iglesias PM, Vegas-Sánchez MC, Torrado-Durán S, Bolás-Fernández F. Torrado-Santiago. Changed crystallinity of mebendazole solid dispersion: improved anthelmintic activity. Int J Pharm. 2011;403(1-2):23–8. https://doi.org/10.1016/j.ijpharm.2010.10.002.
Higuchi T, Connors K. Phase-solubility techniques. Adv Anal Chem Instrum. 1965;4:117–23.
Reyes-Gasga J, Martinez-Pineiro EL, Rodriguez-Alvarez G, Tiznado-Orozco GE, Garcia-Garcia R, Bres EF. XRD and FTIR crystallinity indices in sound human tooth enamel and synthetic hydroxyapatite. Mater Sci Eng C. 2013;33(8):4568–74.
Grimaudo MA, Pescina S, Padula C, Santi P, Concheiro A, Alvarez-Lorenzo C, et al. Poloxamer 407/TPGS mixed micelles as promising carriers for cyclosporine ocular delivery. Mol Pharm. 2018;15(2):571–84. https://doi.org/10.1021/acs.molpharmaceut.7b00939.
Suksiriworapong J, Rungvimolsin T, A-gomol A, Junyaprasert VB, Chantasart D. Development and characterization of lyophilized diazepam loaded polymeric micelles. AAPS PharmSciTech. 2014;15(1):52–64. https://doi.org/10.1208/s12249-013-0032-4.
Dos Santos KM, Barbosa RM, Vargas FGA, de Azevedo EP, Lins ACDS, Camara CA, et al. Development of solid dispersions of β-lapachone in PEG and PVP by solvent evaporation method. Drug Dev Ind Pharm. 2018;44(5):750–6. https://doi.org/10.1080/03639045.2017.1411942.
Tothadi S, Bhogala BR, Gorantla AR, Thakur TS, Jetti RK, Desiraju GR. Triclabendazole: an intriguing case of co-existence of conformational and tautomeric polymorphism. Chemistry Asian J. 2012;7(2):330–42. https://doi.org/10.1002/asia.201100638.
Ali W, Williams AC, Rawlinson CF. Stochiometrically governed molecular interactions in drug: poloxamer solid dispersions. Int J Pharm. 2010;391(1-2):162–8. https://doi.org/10.1016/j.ijpharm.2010.03.014.
Xiang TX, Anderson BD. Effects of molecular interactions on miscibility and mobility of ibuprofen in amorphous solid dispersions with various polymers. J Pharm Sci. 2019;108(1):178–86. https://doi.org/10.1016/j.xphs.2018.10.052.
Truong DH, Tran TH, Ramasamy T, Choi JY, Choi H, Yong CS, et al. Preparation and characterization of solid dispersion using a novel amphiphilic copolymer to enhance dissolution and oral bioavailability of sorafenib. Powder Technol. 2015;283:260–5. https://doi.org/10.1016/j.powtec.2015.04.044.
Weuts I, Van Dycke F, Voorspoels J, De Cort S, Stokbroekx S, Leemans R, et al. Physicochemical properties of 720 the amorphous drug, cast films, and spray dried powders to predict formulation probability of success for solid dispersions: etravirine. J Pharm Sci. 2011;100(1):260–74. https://doi.org/10.1002/jps.22242.
Bley H, Fussnegger B, Bodmeier R. Characterization and stability of solid dispersions based on PEG/polymer blends. Int J Pharm. 2010;390(2):165–73. https://doi.org/10.1016/j.ijpharm.2010.01.039.
Anand O, Yu LX, Conner DP, Davit BM. Dissolution testing for generic drugs: an FDA perspective. AAPS J. 2011;13(3):328–35. https://doi.org/10.1208/s12248-011-9272-y.
EMEA. European Medicines Agency. Note for guidance on the investigation of bioavailability and bioequivalence, vol. 1. London: European Medicines Agency; 2001. 18p
FDA. Food and Drug Administration. Guidance for industry: dissolution testing of immediate release solid oral dosage forms, vol. 1. Rockville: Food and Drug Administration; 1997. 11p
Wu C, Liu Y, He Z, Sun J. Insight into the development of dissolution media for BCS class II drugs: a review from quality control and prediction of in vivo performance perspectives. Curr Drug Deliv. 2016;13(7):1004–20.
Bull World Health Organ 96. 2018. pp. 378–385.
Chutimaworapan S, Ritthidej GC, Yonemochi E, Oguchi T, Yamamoto K. Effect of water-soluble carriers on dissolution characteristics of nifedipine solid dispersions. Drug Dev Ind Pharm. 2000;26(11):1141–50. https://doi.org/10.1081/DDC-100100985.
Guncheva G, Stippler E. Effect of four commonly used dissolution media surfactants on pancreatin proteolytic activity. AAPS PharmSciTech. 2017;18(4):1402–7. https://doi.org/10.1208/s12249-016-0618-8.
von Orelli J, Leuenberger H. Search for technological reasons to develop a capsule or a tablet formulation with respect to wettability and dissolution. Int J Pharm. 2004;287(1-2):135–45. https://doi.org/10.1016/j.ijpharm.2004.09.006.
Nair R, Vemuri M, Agrawala P, Kim S. Investigation of various factors affecting encapsulation on the in-cap automatic capsule-filling machine. AAPS PharmSciTech. 2004;5(4):46–53. https://doi.org/10.1208/pt050457.
Leonardi D, Salomon CJ. Unexpected performance of physical mixtures over solid dispersions on the dissolution behavior of benznidazole from tablets. J Pharm Sci. 2013;102(3):1016–23. https://doi.org/10.1002/jps.23448.
Shah RB, Tawakkul MA, Khan MA. Comparative evaluation of flow for pharmaceutical powders and granules. AAPS PharmSciTech. 2008;9(1):250–8. https://doi.org/10.1208/s12249-008-9046-8.
Emery E, Oliver J, Pugsley T, Sharma J, Zhou J. Flowability of moist pharmaceutical powders. Powder Technol. 2009;189(3):409–15. https://doi.org/10.1016/j.powtec.2008.06.017.
The United States Pharmacopeial Convention, Rockville: USP 42 NF 37: United States Pharmacopeia Convention; 2019.
Karataş A, Bekmezci S. Evaluation and enhancement of physical stability of semi-solid dispersions containing piroxicam into hard gelatin capsules. Acta Pol Pharm. 2013;70(5):883–97.
Bahl D, Bogner RH. Amorphization of indomethacin by co-grinding with Neusilin US2: amorphization kinetics, physical stability and mechanism. Pharm Res. 2006;23(10):2317–25. https://doi.org/10.1007/s11095-006-9062-x.
Acknowledgments
D.R. thanks CONICET for a Ph.D. fellowship.
Funding
The authors of this study received financial support from MINCYT (Argentina), CONICET (Argentina), and National University of Rosario (Argentina).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Real, D., Orzan, L., Leonardi, D. et al. Improving the Dissolution of Triclabendazole from Stable Crystalline Solid Dispersions Formulated for Oral Delivery. AAPS PharmSciTech 21, 16 (2020). https://doi.org/10.1208/s12249-019-1551-4
Received:
Accepted:
Published:
DOI: https://doi.org/10.1208/s12249-019-1551-4