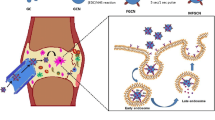
Abstract
Etanercept (ETA) has been used as a drug to neutralise tumour necrotic factor alpha (TNF-α) for treatment of rheumatoid arthritis (RA), yet there are limitations concerning its low specific drug targeting and side effects. In this study, agarose–curdlan encapsulating etanercept (ACE) gel was successfully formulated and evenly distributed as nano-particles of 30–100 nm diameter, exhibiting an ETA encapsulation efficiency of 73.8% and an ETA-releasing efficiency of 50% after 52 h. The number of dectin-1-overexpressing macrophage cells, RAW264.7, exposed to ACE that migrated in the Boyden chamber assay was equal to that exposed to either agarose–curdlan or curdlan nanogels, but substantially higher than those exposed to agarose gel and water-soluble ETA by 67 and 141 fold, respectively (p < 0.05), suggesting the targeting effect of curdlan on dectin-1. Enzyme-linked immunosorbent assay revealed that the ETA released from the ACE nanogel could neutralise TNF-α secreted by lipopolysaccharide (LPS)-induced RAW264.7. Moreover, at 24 h and 72 h, the released ETA showed 1.3- to 4.4-fold greater effectiveness, respectively, than water-soluble ETA. This study demonstrates that the ACE nanogel can attract immune cells and slowly release ETA to efficiently neutralise the TNF-α produced by these cells and, thus, could be a promising ETA carrier for targeted RA treatment.
Similar content being viewed by others
References
D.S. Dimitrov, Therapeutic Proteins (Berlin: Springer, 2012), pp. 1–26.
T.S. Plantinga, J. Fransen, N. Takahashi, R. Stienstra, P.L. van Riel, W.B. van den Berg, M.G. Netea, and L.A. Joosten, Arthritis Res. Ther. 12, R26 (2010).
L.M. van den Berg, E.M. Zijlstra-Willems, C.D. Richters, M.M. Ulrich, and T.B. Geijtenbeek, Cell. Immunol. 289, 49 (2014).
E.H. Choy and G.S. Panayi, N. Engl. J. Med. 344, 907 (2001).
C. Tetta, G. Camussi, V. Modena, C. Di Vittorio, and C. Baglioni, Ann. Rheum. Dis. 49, 665 (1990).
H. Matsuno, K. Yudoh, R. Katayama, F. Nakazawa, M. Uzuki, T. Sawai, T. Yonezawa, Y. Saeki, G.S. Panayi, and C. Pitzalis, Rheumatology 41, 329 (2002).
Y.-F. Chen, P. Jobanputra, P. Barton, S. Jowett, S. Bryan, W. Clark, A. Fry-Smith, and A. Burls, Health Technol. Assess. 10, 1 (2006).
Z. Kaymakcalan, P. Sakorafas, S. Bose, S. Scesney, L. Xiong, D.K. Hanzatian, J. Salfeld, and E.H. Sasso, Clin. Immunol. 131, 308 (2009).
K. Malottki, P. Barton, A. Tsourapas, A.O. Uthman, Z. Liu, K. Routh, M. Connock, P. Jobanputra, D. Moore, A. Fry-Smith, and Y.-F. Chen, Health Technol. Assess. 15, 1 (2011).
U.G. Longo, S. Petrillo, and V. Denaro, Int. J. Rheumatol. 2015, 648073 (2015).
J. Panyam and V. Labhasetwar, Adv. Drug Deliv. Rev. 55, 329 (2003).
N. Wang and X.S. Wu, Int. J. Pharm. 166, 1 (1998).
N. Wang and X.S. Wu, Pharm. Dev. Technol. 2, 135 (1997).
P. Serwer, Agarose gels: properties and use for electrophoresis. Electrophoresis 4, 375 (1983).
P. Zarrintaj, S. Manouchehri, Z. Ahmadi, M.R. Saeb, A.M. Urbanska, D.L. Kaplan, and M. Mozafari, Carbohydr. Polym. 187, 66 (2018).
J. Liu, S. Lin, L. Li, and E. Liu, Int. J. Pharm. 298, 117 (2005).
O. Erdemli, S. Ozen, D. Keskin, A. Usanmaz, E.D. Batu, B. Atilla, and A. Tezcaner, J. Biomater. Appl. 29, 524 (2014).
M. Giulbudagian, G. Yealland, S. Hönzke, A. Edlich, B. Geisendörfer, B. Kleuser, S. Hedtrich, and M. Calderón, Theranostics 8, 450 (2018).
K.B. Soo, J.I. Duck, K.J. Sik, L. Jung-heon, L.I. Young, and L.K. Bok, Biotechnol. Lett. 22, 1127 (2000).
M. Kanke, K. Koda, Y. Koda, and H. Katayama, Pharm. Res. 9, 414 (1992).
Z. Cai and H. Zhang, Food Hydrocoll. 68, 128 (2017).
Y. Matsumura and H. Maeda, Cancer Res. 46, 6387 (1986).
X. Ma, Y. Xia, L. Ni, L. Song, and Z. Wang, Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 121, 657 (2014).
E.J. Lee, J.K. Park, S.A. Khan, and K.H. Lim, J. Chem. Eng. Jpn 44, 502 (2011).
B.G. Zanetti-Ramos, E. Lemos-Senna, V. Soldi, R. Borsali, E. Cloutet, and H. Cramail, Polymer 47, 8080 (2006).
D. Lu and A.J. Hickey, AAPS PharmSciTech 6, E641 (2005).
H.C. Chen, Cell Migration (Berlin: Springer, 2005), pp. 15–22.
W. Hergert, Chapter 2. Springer Series in Optical Sciences, Vol. 169 (Berlin: Springer, 2012).
E. Al-Emam, A.G. Motawea, K. Janssens, and J. Caen, Herit. Sci. 7, 22 (2019).
Y. Adachi, T. Ishii, Y. Ikeda, A. Hoshino, H. Tamura, J. Aketagawa, S. Tanaka, and N. Ohno, Infect. Immun. 72, 4159 (2004).
A.S. Palma, T. Feizi, Y. Zhang, M.S. Stoll, A.M. Lawson, E. Díaz-Rodríguez, M.A. Campanero-Rhodes, J. Costa, S. Gordon, and G.D. Brown, J. Biol. Chem. 281, 5771 (2006).
D.A. Salvatore, P. Carlo, C. Fabrizio, L. Ennio, M. Antonio, M. Alessandro, S. Carlo, S. Raffaele, S. Antonio, and O. Ignazio, Clin. Exp. Rheumatol. 29, 865 (2011).
X. Chen, D.C. DuBois, R.R. Almon, and W.J. Jusko, Drug Metab. Dispos. 43, 898 (2015).
O.C. Farokhzad and R. Langer, ACS Nano 3, 16 (2009).
M. Moscovici, C. Hlevca, C. Angela, and R.D. Pavaloiu, Nanocellulose and Nanohydrogel Matrices: Biotechnological and Biomedical Applications (Colorado: Wiley, 2017), pp. 209–269.
H. Schierbeck, H. Wähämaa, U. Andersson, and H.E. Harris, Mol. Med. 16, 343 (2010).
J.H. Egberts, V. Cloosters, A. Noack, B. Schniewind, L. Thon, S. Klose, B. Kettler, C. von Forstner, C. Kneitz, and J. Tepel, Can. Res. 68, 1443 (2008).
S.H. Zhao, X.T. Wu, W.C. Guo, Y.M. Du, L. Yu, and J. Tang, Int. J. Pharm. 393, 269 (2010).
J. Varshosaz, M.R. Zaki, M. Minaiyan, and J. Banoozadeh, Biomed. Res. Int. 2015, 571816 (2015).
Z. Hu, S. Li, and L. Yang, Polímeros 22, 422 (2012).
Y. Xu, Y. Du, R. Huang, and L. Gao, Biomaterials 24, 5015 (2003).
J. Han, J. Cai, W. Borjihan, T. Ganbold, T.M. Rana, and H. Baigude, Carbohydr. Polym. 117, 324 (2015).
S. Mochizuki and K. Sakurai, J. Control. Release 151, 155 (2011).
Acknowledgement
This work was supported by a grant from Vietnam National University, Hanoi to Huong T. T.Pham Under project number KLEPT.16.01. We thank Msc. Phan Thi Kieu Trang from the Faculty of Environmental Engineering, University of Kitakyushu for her kind guidance on setting the drug targeting experiment, and Dr. Pham Bao Yen from the Key Laboratory of Enzyme and Protein Technology, VNU University of Science, Vietnam National University-Hanoi for English correction
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Nguyen, D.H., Nguyen, N.B., Nguyen, L.T. et al. An Agarose–Curdlan Nanogel that Carries Etanercept to Target and Neutralises TNF-α Produced by Dectin-1-Expressing Immune Cells. J. Electron. Mater. 48, 6570–6582 (2019). https://doi.org/10.1007/s11664-019-07458-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-019-07458-2