Abstract
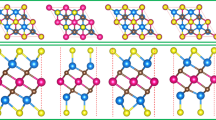
In this work, we present a study on the atomic details and the electronic structure of the NiCo2S4/KOH electrolyte interface via first-principles calculation. Both Ni-terminated and CoS2-terminated NiCo2S4 (001) surfaces are studied in order to understand the electronic structure of the interface and surface reactivity. The surface free energy indicates that the Ni-capped NiCo2S4 (001) surface is more stable than the CoS2-capped one. Upon adsorption, KOH received more electrons (0.1e) from the latter surface than from the former and the planar average differential charge density indicates that a larger electric dipole forms in CoS2-terminated NiCo2S4/KOH interface. These analyses prove that the CoS2-terminated NiCo2S4 (001) surface presents a stronger affinity for OH− in the electrolyte, which leads to the enhanced electrochemical performance. These results will not only contribute to a better understanding of the fundamental knowledge of the NiCo2S4/KOH electrolyte interface, but also provide a guidance as to enhance the electrochemical activity of NiCo2S4 materials, which will benefit the ultimate application of NiCo2S4 (001) as electrode material for electrochemical energy storage.
Similar content being viewed by others
References
C.Z. Wei, N.N. Zhan, J. Tao, S.S. Pang, L.P. Zhang, C. Cheng, and D.J. Zhang, Appl. Surf. Sci. 453, 288 (2018).
C. Liu and X. Wu, Mater. Res. Bull. 103, 55 (2018).
T. Xiao, J. Li, X.Y. Zhuang, W. Zhang, S.L. Wang, X.L. Chen, P. Xiang, L.H. Jiang, and X.Y. Tan, Electrochim. Acta 269, 397 (2018).
J. Yang, C. Yu, X.M. Fan, S.X. Liang, S.F. Li, H.W. Huang, Z. Ling, C. Hao, and J.S. Qiu, Energy Environ. Sci. 9, 1299 (2016).
T.B. He, S.L. Wang, F.X. Lu, M.C. Zhang, X. Zhang, and L. Xu, RSC Adv. 6, 97352 (2016).
H.C. Chen, J.J. Jiang, L. Zhang, D.D. Xia, Y.D. Zhao, D.Q. Guo, T. Qi, and H.Z. Wan, J. Power Sources 254, 249 (2014).
H.Z. Wan, J.J. Jiang, J.W. Yu, K. Xu, L. Miao, L. Zhang, H.C. Chen, and Y.J. Ruan, CrystEngComm 15, 7649 (2013).
Y.X. Wen, S.L. Peng, Z.L. Wang, J.X. Hao, T.F. Qin, S.Q. Lu, J.C. Zhang, D.Y. He, X.Y. Fan, and G.Z. Cao, J. Mater. Chem. A 5, 7144 (2017).
X.H. Lu, M.H. Yu, G.M. Wang, Y.X. Tong, and Y. Li, Energy Environ. Sci. 7, 2160 (2014).
Z.Q. Chai, Z.X. Wang, J.X. Wang, X.H. Li, and H.J. Guo, Ceram. Int. 44, 15778 (2018).
Y.S. Wu, X.J. Liu, D.D. Han, X.Y. Song, L. Shi, Y. Song, S.W. Niu, Y.F. Xie, J.Y. Cai, and S.Y. Wu, Nat. Commun. 9, 1425 (2018).
J. Liu, J.S. Wang, B. Zhang, Y.J. Ruan, L. Lv, X. Ji, K. Xu, L. Miao, and J.J. Jiang, ACS Appl. Mater. Interfaces 9, 15364 (2017).
D.N. Liu, Q. Lu, Y.L. Luo, X.P. Sun, and A.M. Asiri, Nanoscale 7, 15122 (2015).
Q. Liu, J.T. Jin, and J.Y. Zhang, ACS Appl. Mater. Interfaces 5, 5002 (2013).
J.Y. Lin and S.W. Chou, Electrochem. Commun. 37, 11 (2013).
J.W. Xiao, X.W. Zeng, W. Chen, F. Xiao, and S. Wang, Chem. Commun. 49, 11734 (2013).
C. Xia, P. Li, A.N. Gandi, U. Schwingenschlogl, and H.N. Alshareef, Chem. Mater. 27, 6482 (2015).
O. Knop, K.I.G. Reid, Sutarno, and Y. Nakagawa, Can. J. Chem. 46, 3463 (1968).
A. Candan and G. Uğur, Mod. Phys. Lett. B 30, 16500020 (2016).
A. Manzar, G. Murtaza, R. Khenata, and S. Muhammad, Chin. Phys. Lett. 30, 127401 (2013).
H.G. Yang, C.H. Sun, S.Z. Qiao, J. Zou, G. Liu, S.C. Smith, H.M. Cheng, and G.Q. Lu, Nature 453, 638 (2008).
H.H. Alkam, K.M. Kanan, and C.C. Hadjikostas, Polyhedron 24, 2944 (2005).
G.D. Gatta, G.J. McIntyre, R. Sassi, N. Rotiroti, and A. Pavese, Am. Mineral. 96, 34 (2010).
W. Carrillo-Cabrera, J. Saßmannshausen, H.G. von Schnering, F. Menzel, and W. Brockner, Z. Anorg. Allg. Chem. 620, 489 (1994).
S. Grimme, J. Comput. Chem. 25, 1463 (2004).
Acknowledgments
This work was supported by the National Natural Science Foundation of China under Grant (Nos. 41076057, 41476082), China Postdoctoral Science Foundation (Grant No. 2017M612348) and project from Ocean University of China (Grant No. 3002000-861701013151).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hu, H.H., Dou, K.P., Yuan, G. et al. Computational Studies on Structural and Electronic Properties of NiCo2S4 (001)/KOH Electrolyte Interface. J. Electron. Mater. 48, 6347–6353 (2019). https://doi.org/10.1007/s11664-019-07423-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11664-019-07423-z