Abstract
Background
Adnexal carcinomas are rare and heterogeneous skin tumors, for which no standard treatments exist for locally advanced or metastatic tumors.
Aim of the study
To evaluate the expression of PD-L1 and CD8 in adnexal carcinomas, and to study the association between PD-L1 expression, intra-tumoral T cell CD8+ infiltrate, and metastatic evolution.
Materials and methods
Eighty-three adnexal carcinomas were included. Immunohistochemistry using anti-PD-L1 monoclonal antibodies (E1L3N and 22C3) and CD8 was performed. PD-L1 expression in tumor and immune cells, and CD8+ tumor-infiltrating lymphocyte (TIL) density were analyzed semi-quantitatively.
Results
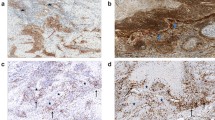
Among the 60 sweat gland, 18 sebaceous and 5 trichoblastic carcinomas, 11% expressed PD-L1 in ≥ 1% tumor cells, more frequently sweat gland carcinomas (13%, 8/60) including apocrine carcinoma (40%, 2/5) and invasive extramammary Paget disease (57%, 4/7). Immune cells expressed significantly more PD-L1 than tumor cells (p < 0.01). Dense CD8+ TILs were present in 60% trichoblastic, 43% sweat gland, and 39% sebaceous carcinomas. CD8+ TILs were associated with PD-L1 expression by tumor cells (p < 0.01). Thirteen patients out of 47 developed metastases (27%) with a median follow-up of 30.5 months (range 7–36). Expression of PD-L1 by tumor cells was associated with the development of metastasis in univariate analysis (HR 4.0, 95% CI 1.1–15, p = 0.0377) but not in multivariate analysis (HR 4.1, 95% CI 0.6–29, p = 0.15).
Conclusion
PD-L1 expression is highly heterogeneous among adnexal carcinoma subtypes, higher in apocrine carcinoma and invasive extramammary Paget disease, and associated with CD8+ TILs. Our data suggest the interest of evaluating anti-PD1 immunotherapy in advanced or metastatic cutaneous adnexal carcinoma.

Similar content being viewed by others
Abbreviations
- AJCC:
-
American Joint Committee on Cancer
- APHP:
-
Assistance Publique-Hôpitaux de Paris
- BCC:
-
Basal cell carcinoma
- CHRU:
-
Centre hospitalier régional universitaire
- cSCC:
-
Cutaneous squamous cell carcinoma
- EMPD:
-
Extramammary Paget disease
- IC:
-
Immune cells
- NOS:
-
No other specification
- TC:
-
Tumor cells
- TMB:
-
Tumor mutation burden
- UMR:
-
Unité mixte de recherche
- WHO:
-
World Health Organization
References
Crowson AN, Magro CM, Mihm MC (2006) Malignant adnexal neoplasms. Mod Pathol 19(Suppl 2):S93–S126
Cardoso JC, Calonje E (2015) Malignant sweat gland tumours: an update. Histopathology 67(5):589–606
Martinez SR, Barr KL, Canter RJ (2011) Rare tumors through the looking glass: an examination of malignant cutaneous adnexal tumors. Arch Dermatol 147(9):1058–1062
Danialan R, Mutyambizi K, Aung P et al (2015) Challenges in the diagnosis of cutaneous adnexal tumours. J Clin Pathol 68(12):992–1002
Suchak R, Wang WL, Prieto VG et al (2012) Cutaneous digital papillary adenocarcinoma: a clinicopathologic study of 31 cases of a rare neoplasm with new observations. Am J Surg Pathol 36(12):1883–1891
Ho VH, Ross MI, Prieto VG et al (2007) Sentinel lymph node biopsy for sebaceous cell carcinoma and melanoma of the ocular adnexa. Arch Otolaryngol Head Neck Surg 133(8):820–826
Snow SN, Reizner GT (1992) Eccrine porocarcinoma of the face. J Am Acad Dermatol 27(2 Pt 2):306–311
Maeda T, Mori H, Matsuo T et al (1996) Malignant eccrine poroma with multiple visceral metastases: report of a case with autopsy findings. J Cutan Pathol 23(6):566–570
Waqas O, Faisal M, Haider I et al (2017) Retrospective study of rare cutaneous malignant adnexal tumors of the head and neck in a tertiary care cancer hospital: a case series. J Med Case Rep 11(1):67
Hibler BP, Barker CA, Hollmann TJ et al (2017) Metastatic cutaneous apocrine carcinoma: multidisciplinary approach achieving complete response with adjuvant chemoradiation. JAAD Case Rep 3(3):259–262
Bernardez C, Requena L (2018) Treatment of malignant cutaneous adnexal neoplasms. Actas Dermosifiliogr 109(1):6–23
Ishida Y, Agata Y, Shibahara K et al (1992) Induced expression of PD-1, a novel member of the immunoglobulin gene superfamily, upon programmed cell death. EMBO J 11(11):3887–3895
Dong H, Strome SE, Salomao DR et al (2002) Tumor-associated B7-H1 promotes T-cell apoptosis: a potential mechanism of immune evasion. Nat Med 8(8):793–800
Wu P, Wu D, Li L et al (2015) PD-L1 and survival in solid tumors: a meta-analysis. PLoS One 10(6):e0131403
Madore J, Strbenac D, Vilain R et al (2016) PD-L1 negative status is associated with lower mutation burden, differential expression of immune-related genes, and worse survival in stage III melanoma. Clin Cancer Res 22(15):3915–3923
Gadiot J, Hooijkaas AI, Kaiser AD et al (2011) Overall survival and PD-L1 expression in metastasized malignant melanoma. Cancer 117(10):2192–2201
Herbst RS, Soria JC, Kowanetz M et al (2014) Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 515(7528):563–567
Dieci MV, Radosevic-Robin N, Fineberg S et al (2018) Update on tumor-infiltrating lymphocytes (TILs) in breast cancer, including recommendations to assess TILs in residual disease after neoadjuvant therapy and in carcinoma in situ: a report of the International Immuno-Oncology Biomarker Working Group on Breast Cancer. Semin Cancer Biol 52:16–25
Hendry S, Salgado R, Gevaert T et al (2017) Assessing tumor-infiltrating lymphocytes in solid tumors: a practical review for pathologists and proposal for a standardized method from the International Immuno-Oncology Biomarkers Working Group: Part 2: TILs in melanoma, gastrointestinal tract carcinomas, non-small cell lung carcinoma and mesothelioma, endometrial and ovarian carcinomas, squamous cell carcinoma of the head and neck, genitourinary carcinomas, and primary brain tumors. Adv Anat Pathol 24(6):311–335
Kwon MJ, Rho YS, Nam ES et al (2018) Clinical implication of programmed death-ligand 1 expression in tonsillar squamous cell carcinoma in association with intratumoral heterogeneity, human papillomavirus, and epithelial-to-mesenchymal transition. Hum Pathol 80:28–39
Kim J, Kim S, Lee HS et al (2018) Prognostic implication of programmed cell death 1 protein and its ligand expressions in endometrial cancer. Gynecol Oncol 149(2):381–387
Lee KS, Kwak Y, Ahn S et al (2017) Prognostic implication of CD274 (PD-L1) protein expression in tumor-infiltrating immune cells for microsatellite unstable and stable colorectal cancer. Cancer Immunol Immunother 66(7):927–939
Mori S, Motoi N, Ninomiya H et al (2017) High expression of programmed cell death 1 ligand 1 in lung adenocarcinoma is a poor prognostic factor particularly in smokers and wild-type epidermal growth-factor receptor cases. Pathol Int 67(1):37–44
Kim C, Kim EK, Jung H et al (2016) Prognostic implications of PD-L1 expression in patients with soft tissue sarcoma. BMC Cancer 16:434
Madore J, Vilain RE, Menzies AM et al (2015) PD-L1 expression in melanoma shows marked heterogeneity within and between patients: implications for anti-PD-1/PD-L1 clinical trials. Pigment Cell Melanoma Res 28(3):245–253
Kaunitz GJ, Cottrell TR, Lilo M et al (2017) Melanoma subtypes demonstrate distinct PD-L1 expression profiles. Lab Investig 97(9):1063–1071
Robert C, Schachter J, Long GV et al (2015) Pembrolizumab versus ipilimumab in advanced melanoma. N Engl J Med 372(26):2521–2532
Robert C, Long GV, Brady B et al (2015) Nivolumab in previously untreated melanoma without BRAF mutation. N Engl J Med 372(4):320–330
Schaper K, Kother B, Hesse K et al (2017) The pattern and clinicopathological correlates of programmed death-ligand 1 expression in cutaneous squamous cell carcinoma. Br J Dermatol 176(5):1354–1356
Chang J, Zhu GA, Cheung C et al (2017) Association between programmed death ligand 1 expression in patients with basal cell carcinomas and the number of treatment modalities. JAMA Dermatol 153(4):285–290
Reddy BY, Miller DM, Tsao H (2017) Somatic driver mutations in melanoma. Cancer 123(S11):2104–2117
Harwood CA, Proby CM, Inman GJ et al (2016) The promise of genomics and the development of targeted therapies for cutaneous squamous cell carcinoma. Acta Derm Venereol 96(1):3–16
Ng PK, Li J, Jeong KJ et al (2018) Systematic functional annotation of somatic mutations in cancer. Cancer Cell 33(3):450–462 e10
Le DT, Durham JN, Smith KN et al (2017) Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 357(6349):409–413
Yarchoan M, Hopkins A, Jaffee EM (2017) Tumor mutational burden and response rate to PD-1 inhibition. N Engl J Med 377(25):2500–2501
Morrison C, Pabla S, Conroy JM et al (2018) Predicting response to checkpoint inhibitors in melanoma beyond PD-L1 and mutational burden. J Immunother Cancer 6(1):32
Besaratinia A, Pfeifer GP (2008) Sunlight ultraviolet irradiation and BRAF V600 mutagenesis in human melanoma. Hum Mutat 29(8):983–991
Piris A, Peng Y, Boussahmain C et al (2014) Cutaneous and mammary apocrine carcinomas have different immunoprofiles. Hum Pathol 45(2):320–326
Mahalingam M, Nguyen LP, Richards JE et al (2010) The diagnostic utility of immunohistochemistry in distinguishing primary skin adnexal carcinomas from metastatic adenocarcinoma to skin: an immunohistochemical reappraisal using cytokeratin 15, nestin, p63, D2-40, and calretinin. Mod Pathol 23(5):713–719
Wick MR, Ockner DM, Mills SE et al (1998) Homologous carcinomas of the breasts, skin, and salivary glands. A histologic and immunohistochemical comparison of ductal mammary carcinoma, ductal sweat gland carcinoma, and salivary duct carcinoma. Am J Clin Pathol 109(1):75–84
Polonia A, Pinto R, Cameselle-Teijeiro JF et al (2017) Prognostic value of stromal tumour infiltrating lymphocytes and programmed cell death-ligand 1 expression in breast cancer. J Clin Pathol 70(10):860–867
Arias-Pulido H, Cimino-Mathews A, Chaher N et al (2018) The combined presence of CD20+ B cells and PD-L1+ tumor-infiltrating lymphocytes in inflammatory breast cancer is prognostic of improved patient outcome. Breast Cancer Res Treat 171(2):273–282
Karpathiou G, Chaleur C, Hathroubi S, Habougit C, Peoc’h M (2018) Expression of CD3, PD-L1 and CTLA-4 in mammary and extramammary Paget disease. Cancer Immunol Immunother 67:1297–1303
Lyford-Pike S, Peng S, Young GD et al (2013) Evidence for a role of the PD-1:PD-L1 pathway in immune resistance of HPV-associated head and neck squamous cell carcinoma. Cancer Res 73(6):1733–1741
Knol AC, Nguyen JM, Pandolfino MC et al (2018) PD-L1 expression by tumor cell lines: a predictive marker in melanoma. Exp Dermatol 27(6):647–655
Kim H, Kwon HJ, Park SY et al (2018) Clinicopathological analysis and prognostic significance of programmed cell death-ligand 1 protein and mRNA expression in non-small cell lung cancer. PLoS One 13(6):e0198634
Li X, Li M, Lian Z et al (2016) Prognostic role of programmed death ligand-1 expression in breast cancer: a systematic review and meta-analysis. Target Oncol 11(6):753–761
Garcia-Diez I, Hernandez-Ruiz E, Andrades E et al (2018) PD-L1 expression is increased in metastasizing squamous cell carcinomas and their metastases. Am J Dermatopathol 40:647–654
Lipson EJ, Vincent JG, Loyo M et al (2013) PD-L1 expression in the Merkel cell carcinoma microenvironment: association with inflammation, Merkel cell polyomavirus and overall survival. Cancer Immunol Res 1(1):54–63
Stanton SE, Adams S, Disis ML (2016) Variation in the incidence and magnitude of tumor-infiltrating lymphocytes in breast cancer subtypes: a systematic review. JAMA Oncol 2(10):1354–1360
Santoiemma PP, Powell DJ Jr (2015) Tumor infiltrating lymphocytes in ovarian cancer. Cancer Biol Ther 16(6):807–820
Behr DS, Peitsch WK, Hametner C et al (2014) Prognostic value of immune cell infiltration, tertiary lymphoid structures and PD-L1 expression in Merkel cell carcinomas. Int J Clin Exp Pathol 7(11):7610–7621
El Sissy C, Marliot F, Haicheur N et al (2017) Focus on the Immunoscore and its potential clinical implications. Ann Pathol 37(1):29–38
Azimi F, Scolyer RA, Rumcheva P et al (2012) Tumor-infiltrating lymphocyte grade is an independent predictor of sentinel lymph node status and survival in patients with cutaneous melanoma. J Clin Oncol 30(21):2678–2683
Thomas NE, Busam KJ, From L et al (2013) Tumor-infiltrating lymphocyte grade in primary melanomas is independently associated with melanoma-specific survival in the population-based genes, environment and melanoma study. J Clin Oncol 31(33):4252–4259
Kakavand H, Vilain RE, Wilmott JS et al (2015) Tumor PD-L1 expression, immune cell correlates and PD-1+ lymphocytes in sentinel lymph node melanoma metastases. Mod Pathol 28(12):1535–1544
Obeid JM, Erdag G, Smolkin ME et al (2016) PD-L1, PD-L2 and PD-1 expression in metastatic melanoma: correlation with tumor-infiltrating immune cells and clinical outcome. Oncoimmunology 5(11):e1235107
Kluger HM, Zito CR, Barr ML et al (2015) Characterization of PD-L1 expression and associated T-cell infiltrates in metastatic melanoma samples from variable anatomic sites. Clin Cancer Res 21(13):3052–3060
Xu F, Feng G, Zhao H et al (2015) Clinicopathologic significance and prognostic value of B7 homolog 1 in gastric cancer: a systematic review and meta-analysis. Medicine (Baltimore) 94(43):e1911
Guo Y, Yu P, Liu Z et al (2016) Prognostic and clinicopathological value of programmed death ligand-1 in breast cancer: a meta-analysis. PLoS One 11(5):e0156323
Xu F, Xu L, Wang Q et al (2015) Clinicopathological and prognostic value of programmed death ligand-1 (PD-L1) in renal cell carcinoma: a meta-analysis. Int J Clin Exp Med 8(9):14595–14603
Gao HL, Liu L, Qi ZH et al (2018) The clinicopathological and prognostic significance of PD-L1 expression in pancreatic cancer: a meta-analysis. Hepatobiliary Pancreat Dis Int 17(2):95–100
Badoual C, Hans S, Merillon N et al (2013) PD-1-expressing tumor-infiltrating T cells are a favorable prognostic biomarker in HPV-associated head and neck cancer. Cancer Res 73(1):128–138
Droeser RA, Hirt C, Viehl CT et al (2013) Clinical impact of programmed cell death ligand 1 expression in colorectal cancer. Eur J Cancer 49(9):2233–2242
Toyokawa G, Takada K, Haratake N et al (2016) Favorable disease-free survival associated with programmed death ligand 1 expression in patients with surgically resected small-cell lung cancer. Anticancer Res 36(8):4329–4336
Gutermuth J, Audring H, Voit C et al (2004) Antitumour activity of paclitaxel and interferon-alpha in a case of metastatic eccrine porocarcinoma. J Eur Acad Dermatol Venereol 18(4):477–479
Mezger J, Remberger K, Schalhorn A et al (1986) Treatment of metastatic sweat gland carcinoma by a four drug combination chemotherapy: response in two cases. Med Oncol Tumor Pharmacother 3(1):29–34
el-Domeiri AA, Brasfield RD, Huvos AG et al (1971) Sweat gland carcinoma: a clinico-pathologic study of 83 patients. Ann Surg 173(2):270–274
De Iuliis F, Amoroso L, Taglieri L et al (2014) Chemotherapy of rare skin adnexal tumors: a review of literature. Anticancer Res 34(10):5263–5268
Sridhar KS, Benedetto P, Otrakji CL et al (1989) Response of eccrine adenocarcinoma to tamoxifen. Cancer 64(2):366–370
Battistella M, Mateus C, Lassau N et al (2010) Sunitinib efficacy in the treatment of metastatic skin adnexal carcinomas: report of two patients with hidradenocarcinoma and trichoblastic carcinoma. J Eur Acad Dermatol Venereol 24(2):199–203
Hidaka T, Fujimura T, Watabe A et al (2012) Successful treatment of HER-2-positive metastatic apocrine carcinoma of the skin with lapatinib and capecitabine. Acta Derm Venereol 92(6):654–655
Acknowledgements
The authors thank the Tumorothèque of Saint Louis hospital for providing tissue material.
Funding
No relevant funding.
Author information
Authors and Affiliations
Contributions
LD designed the study, acquired and analyzed data, drafted the work and revised it. AO acquired data and revised the work for important intellectual content. BC analyzed data and revised the work for important intellectual content. LM analyzed data and revised the work for important intellectual content. ADM acquired and analyzed data, did the statistical analysis, and revised the work for important intellectual content. NB-S acquired clinical data and revised the work for important intellectual content. CL acquired clinical data and revised the work for important intellectual content. MB designed the study, acquired and analyzed data, drafted the work, and revised the work for important intellectual content. All authors approved the final manuscript version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Informed consent
All patients were informed that part of the remaining tissue material could be used for research, and gave their consent according to the Helsinki declaration. Samples were obtained from the Tumorothèque of Saint-Louis Hospital (Tumor bank registration number DC2009.929, Ministry of Health, France).
Ethical standards
According to the bioethics French law of August 6th 2004, applicable at the time of the study, in the context of a retrospective monocentric non-interventional study, additional ethical committee approval was not necessary.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Duverger, L., Osio, A., Cribier, B. et al. Heterogeneity of PD-L1 expression and CD8 tumor-infiltrating lymphocytes among subtypes of cutaneous adnexal carcinomas. Cancer Immunol Immunother 68, 951–960 (2019). https://doi.org/10.1007/s00262-019-02334-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00262-019-02334-8