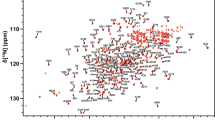
Abstract
The ever-increasing occurrence of antibiotic resistance presents a major threat to public health. Specifically, resistance conferred by β-lactamases places the efficacy of currently available antibiotics at risk. Klebsiella pneumoniae carbapenemase-2 (KPC-2) is a β-lactamase that enables carbapenem resistance and represents a clear and present danger to global public health. In order to combat bacterial infections harboring KPC-2 expression, inhibitors with improved potency need to be developed. Although the structure of KPC-2 has been solved by X-ray crystallography, NMR provides the unique opportunity to study the structure and dynamics of flexible loop regions in solution. Here we report the 1H, 13C, and 15N backbone chemical shift assignments for KPC-2 in the apo state as the first step towards the study of KPC-2 dynamics in the presence and absence of ligands to enable the rational design of optimized inhibitors.


Similar content being viewed by others
References
Barnes MD, Winkler ML, Taracila MA et al (2017) Klebsiella pneumoniae Carbapenemase-2 (KPC-2), Substitutions at Ambler position Asp179, and resistance to ceftazidime-avibactam: unique antibiotic-resistant phenotypes emerge from β-lactamase protein engineering. MBio 8:e00528–e00517. https://doi.org/10.1128/mBio.00528-17
Blair JMA, Webber MA, Baylay AJ et al (2015) Molecular mechanisms of antibiotic resistance. Nat Rev Micro 13:42–51. https://doi.org/10.1038/nrmicro3380
Coleman K (2011) Diazabicyclooctanes (DBOs): a potent new class of non-β-lactam β-lactamase inhibitors. Curr Opin Microbiol 14:550–555. https://doi.org/10.1016/j.mib.2011.07.026
Delaglio F, Grzesiek S, Vuister GW et al (1995) NMRPipe: a multidimensional spectral processing system based on UNIX pipes. J Biomol NMR 6:277–293
Ehmann DE, Jahić H, Ross PL et al (2013) Kinetics of Avibactam Inhibition against Class A, C, and D β-Lactamases. J Biol Chem 288:27960–27971. https://doi.org/10.1074/jbc.M113.485979
Ke W, Bethel CR, Thomson JM et al (2007) Crystal structure of KPC-2: insights into carbapenemase activity in class A beta-lactamases. Biochemistry 46:5732–5740. https://doi.org/10.1021/bi700300u
Krishnan NP, Nguyen NQ, Papp-Wallace KM et al (2015) Inhibition of Klebsiella β-Lactamases (SHV-1 and KPC-2) by Avibactam: a structural study. PLoS ONE 10:e0136813. https://doi.org/10.1371/journal.pone.0136813
Lee W, Tonelli M, Markley JL (2015) NMRFAM-SPARKY: enhanced software for biomolecular NMR spectroscopy. Bioinformatics 31:1325–1327. https://doi.org/10.1093/bioinformatics/btu830
Levitt PS, Papp-Wallace KM, Taracila MA et al (2012) Exploring the role of a conserved class A residue in the Ω-Loop of KPC-2 β-lactamase: a mechanism for ceftazidime hydrolysis. J Biol Chem 287:31783–31793. https://doi.org/10.1074/jbc.M112.348540
Majiduddin FK, Materon IC, Palzkill TG (2002) Molecular analysis of beta-lactamase structure and function. Int J Med Microbiol 292:127–137. https://doi.org/10.1078/1438-4221-00198
Munoz-Price LS, Poirel L, Bonomo RA et al (2013) Clinical epidemiology of the global expansion of Klebsiella pneumoniae carbapenemases. Lancet Infect Dis 13:785–796. https://doi.org/10.1016/S1473-3099(13)70190-7
Nordmann P, Cuzon G, Naas T (2009) The real threat of Klebsiella pneumoniae carbapenemase-producing bacteria. Lancet Infect Dis 9:228–236. https://doi.org/10.1016/S1473-3099(09)70054-4
Palzkill T, Le QQ, Venkatachalam KV et al (1994) Evolution of antibiotic resistance: several different amino acid substitutions in an active site loop alter the substrate profile of beta-lactamase. Mol Microbiol 12:217–229
Papp-Wallace KM, Bethel CR, Distler AM et al (2010) Inhibitor resistance in the KPC-2 beta-lactamase, a preeminent property of this class A beta-lactamase. Antimicrob Agents Chemother 54:890–897. https://doi.org/10.1128/AAC.00693-09
Papp-Wallace KM, Winkler ML, Taracila MA, Bonomo RA (2015) Variants of β-lactamase KPC-2 that are resistant to inhibition by avibactam. Antimicrob Agents Chemother 59:3710–3717. https://doi.org/10.1128/AAC.04406-14
Raquet X, Lamotte-Brasseur J, Fonzé E et al (1994) TEM beta-lactamase mutants hydrolysing third-generation cephalosporins. A kinetic and molecular modelling analysis. J Mol Biol 244:625–639. https://doi.org/10.1006/jmbi.1994.1756
Shapiro AB (2017) Kinetics of Sulbactam Hydrolysis by β-Lactamases, and Kinetics of β-Lactamase Inhibition by Sulbactam. Antimicrob Agents Chemother 61:e01612–e01617. https://doi.org/10.1128/AAC.01612-17
Shen Y, Delaglio F, Cornilescu G, Bax A (2009) TALOS+: a hybrid method for predicting protein backbone torsion angles from NMR chemical shifts. J Biomol NMR 44:213–223. https://doi.org/10.1007/s10858-009-9333-z
Acknowledgements
The authors acknowledge financial support from the National Institutes of Health through the National Institute of General Medical Sciences under Award Number R35GM128595 to RCP and through the National Institute of Allergy and Infectious Diseases under Award Numbers R01AI100560, R01AI063517, R21AI114508, and R01AI072219 to RAB. This study was supported in part by funds and/or facilities provided by the Cleveland Department of Veterans Affairs, Award Number 1I01BX001974 to RAB from the Biomedical Laboratory Research & Development Service of the VA Office of Research and Development and the Geriatric Research Education and Clinical Center VISN 10 to RAB. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the Department of Veterans Affairs. MALDI-TOF data were collected using instrumentation purchased with NSF grant award CHE0839233. The authors acknowledge institutional support from Miami University through the Robert H. and Nancy J. Blayney Professorship to RCP.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
The corresponding author (RCP) is the recipient of a research contract from Allecra Therapeutics to study KPC-2. The authors RCP and RAB are the co-recipients of a research contract (MISP #57394) from Merck & Co. to study inhibition of KPC-2.
Rights and permissions
About this article
Cite this article
VanPelt, J., Shurina, B.A., Ramelot, T.A. et al. 1H, 13C, and 15N backbone resonance assignments for KPC-2, a class A serine-β-lactamase. Biomol NMR Assign 13, 139–142 (2019). https://doi.org/10.1007/s12104-018-9866-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12104-018-9866-8