Abstract
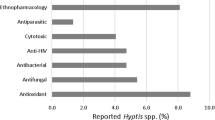
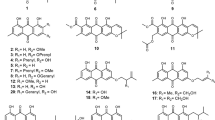
The Stereocaulon genus is one of the fruticose lichen groups distributed worldwide from tropical zones to polar zones. However, the scientific study of this tricky genus is still limited, making it a challenge to study the group further. Detailed morphological studies are essential to discriminate closely shaped species which is illustrated through personal data focused on phyllocladia, apothecia and spores of nine species. Secondary metabolites isolated from Stereocaulon species are mostly some depsides, depsidones, diphenylethers and dibenzofurans which can have a taxonomic value. The use of Stereocaulon lichens as a traditional medicine in several regions of the world and pharmacological studies of extracts and isolated compounds have been compiled. Biological activities as cytotoxic, anti-inflammatory, antibacterial, antifungal or antioxidant are reported.
















Similar content being viewed by others
References
Aghoramurthy K, Sarma K, Seshadri T (1961) Chemical investigation of Indian lichens: part XXV—chemical components of some rare Himalayan lichens. J Sci Ind Res 20B:166–168
Asahina Y, Nonomura S (1935) Untersuchungen über flechtenstoffe, LVI.Mitteil.: Über die konstitution der lobarsäure (I. Mitteil.). Ber Dtsch Chem Ges 68:1698–1704
Baron M, Gorin PA, Iacomini M (1988) Isolation and identification of a linear (1 → 3)-linked β-d-glucan and other carbohydrate components of the lichen Stereocaulon ramulosum (SW.) Räusch. Carbohydr Res 177:235–239. https://doi.org/10.1016/0008-6215(88)85057-2
Baron M, Gorin PAJ, Iacomini M (1989) Structural studies on a galactomannan isolated from the lichen Stereocaulon ramulosum. Agric Biol Chem 53:1751–1758. https://doi.org/10.1080/00021369.1989.10869550
Bhattarai HD, Kim T, Oh H, Yim JH (2013) A new pseudodepsidone from the Antarctic lichen Stereocaulon alpinum and its antioxidant, antibacterial activity. J Antibiot (Tokyo) 66:559–561. https://doi.org/10.1038/ja.2013.41
Boekhout T (1982) Studies on Colombia cryptogams XVIII. the genus Stereocaulon (SCHREBER)HOFFMAN (Lichenes). J Hattori Bot Lab 53:483–511
Bolognese A, Chioccara F, Scherillo G (1974) Isolation and characterization of atranorin and 4,6 dihydroxy-2-methoxy-3-methylacetophenone from Stereocaulon vesuvianum. Phytochemistry 13:1989–1990. https://doi.org/10.1016/0031-9422(74)85131-9
Bruun T (1973) Bourgeanic acid in the lichen Stereocaulon tomentosum. Acta Chem Scand 27:3120
Bruun T (1976) Brassicasterol in Cladonia gonecha and Stereocaulon tomentosum. Phytochemistry 15:1179–1180. https://doi.org/10.1016/0031-9422(76)85128-X
Caccamese S, Compagnini A, Toscano RM, Cascio O (1986) Methyl β-orcinolcarboxylate and atranol from the lichen Stereocaulon vesuvianum. J Nat Prod 49:1159–1160. https://doi.org/10.1021/np50048a049
Cai Z, Song M, Zhang L et al (2009) Phenolic constituents of Stereocaulon paschale Hoffm. Sanxia Daxue Xuebao Ziran Kexueban 31:94–109
Carpentier C, Queiroz EF, Marcourt L et al (2017) Dibenzofurans and pseudodepsidones from the lichen Stereocaulon paschale collected in northern Quebec. J Nat Prod 80:210–214. https://doi.org/10.1021/acs.jnatprod.6b00831
Chooi Y (2008) Genetic potential of lichen-forming fungi in polyketide biosynthesis. RMIT University, Melbourne
Dodge C (1929) A synopsis of Stereocaulon with notes on some exotic species. Ann Cryptog Exot 2:93–153
Duvigneaud P (1942) Contribution à l’étude systématique et chimique du genre Stereocaulon. Biol Jaarb “Dodonaea” 25:80–98
Duvigneaud P (1944) Remarques sur la systématique des lichens à podétions. Bull Jard Bot Bruxelles 17:149–155
Duvigneaud P (1955) Les Stereocaulons des hautes montagnes du Kivu. Rev Bot 14:1–141
Elix J (1996) Biochemistry and secondary metabolites. In: Nash T (ed) Lichen biology. Cambridge University Press, Cambridge, pp 154–180
Elix J (2014) A Catalogue of standardized chromatographic data and biosynthetic relationships for lichen substances, 3rd edn. Canberra
Elix JA, Wardlaw JH (2000) Lusitanic acid, peristictic acid and verrucigeric acid. Three new β-orcinol depsidones from the lichens Relicina sydneyensis and Xanthoparmelia verrucigera. Aust J Chem 53:815–818
Foo LY, Galloway DJ (1979) Pseudodepsidones and other constituents from Xanthoparmelia scabrosa. Phytochemistry 18:1977–1980. https://doi.org/10.1016/S0031-9422(00)82715-6
Fox CH, Huneck S (1970) Inhaltsstoffe von einigen Stereocaulon-arten. Phytochemistry 9:2057. https://doi.org/10.1016/S0031-9422(00)85361-3
Fox CH, Klein E, Huneck S (1970) Colensionsäure, ein neues depsidon aus Stereocaulon colensoi. Phytochemistry 9:2567–2571. https://doi.org/10.1016/S0031-9422(00)85779-9
Fraser M-H, Cuerrier A, Haddad PS et al (2007) Medicinal plants of Cree communities (Québec, Canada): antioxidant activity of plants used to treat type 2 diabetes symptoms. Can J Physiol Pharmacol 85:1200–1214. https://doi.org/10.1139/Y07-108
Goel M, Sharma PK, Dureja P et al (2011) Antifungal activity of extracts of the lichens Parmelia reticulata, Ramalina roesleri, Usnea longissima and Stereocaulon himalayense. Arch Phytopathol Plant Prot 44:1300–1311. https://doi.org/10.1080/03235408.2010.496549
Gonzalez A, Rodriguez E, Bermejo J (1995) Depsidone chemical transformations in an extract of the lichen Stereocaulon azoreum. An Quim 5:461–466
González A, Pérez E, Padrón C, Barrera J (1992) Chemical constituents of the lichen Stereocaulon azoreum. Z Naturforsch C Bio Sci 47:503
Gorin PAJ, Iacomini M (1985) Structural diversity of d-galacto-d-mannan components isolated from lichens having ascomycetous mycosymbionts. Carbohydr Res 142:253–267. https://doi.org/10.1016/0008-6215(85)85027-8
Gupta VK, Darokar MP, Saikia D et al (2007) Antimycobacterial activity of lichens. Pharm Biol 45:200–204. https://doi.org/10.1080/13880200701213088
Hamada N, Ueno T (1990) Lecanoric acid from the mycobiont of the lichen Stereocaulon curtatum. Phytochemistry 29:678–679. https://doi.org/10.1016/0031-9422(90)85147-8
Han YJ, Chan K I, Kyu KD et al (2012) Lobarin for treating diabetes and obesity. Republic of Korea: KIPRIS. 1020100097678
Hauan E, Kjolberg O (1971) Studies on the polysaccharides of lichens. I. The structure of a water–insoluble polysaccharide in Stereocaulon paschale (L.) Fr. Acta Chem Scand 25:2622–2628
Hickey B, Lumsden A, Cole A, Walker J (1990) Antibiotic compounds from New Zealand plants: methyl haematommate, an anti-fungal agent from Stereocaulon ramulosum. NZ Nat Sci 17:49–53
Högnabba F (2006) Molecular phylogeny of the genus Stereocaulon (Stereocaulaceae, lichenized ascomycetes). Mycol Res 110:1080–1092. https://doi.org/10.1016/j.mycres.2006.04.013
Hognabba F, Pino-Bodas R, Nordin A et al (2014) Phylogenetic position of the crustose Stereocaulon species. Lichenol 46:103–114. https://doi.org/10.1017/S002428291300073X
Huneck S (1972) Die sekundärstoffe einiger flechten. Phytochemistry 11:1493–1495. https://doi.org/10.1016/S0031-9422(00)90110-9
Huneck S (1974) Sekundärstoffe einiger Stereocaulon-arten. Phytochemistry 13:2313–2314. https://doi.org/10.1016/0031-9422(74)85046-6
Huneck S, Follman G (1965) Über die inhaltstoffe von Nephroma gyelnikii (RAES) LAMB, Byssocaulon niveum MONT. und Stereocaulon corticulatum NYL. var.procerum LAMB. Z Naturforsch C Bio Sci 20b:1012–1013
Huneck S, Yoshimura I (1996) Identification of lichen substances. Springer, Berlin
Huneck S, Schmidt J, Tabacchi R (1989) Thermal decomposition of lichen depsides. Z Naturforsch C Bio Sci 44:1283. https://doi.org/10.1515/znb-1989-1023
Hylands PJ, Ingolfsdottir K (1985) The isolation of methyl β-orsellinate from Stereocaulon alpinum and comments on the isolation of 4,6-dihydroxy-2-methoxy-3-methylacetophenone from Stereocaulon species. Phytochemistry 24:127–129. https://doi.org/10.1016/S0031-9422(00)80821-3
Index Fungorum (2018) Stereocaulon. http://www.indexfungorum.org/names/Names.asp. Accessed 23 Apr 2018
Ingolfsdottir K, Bloomfield SF, Hylands PJ (1985) In vitro evaluation of the antimicrobial activity of lichen metabolites as potential preservatives. Antimicrob Agents Chemother 28:289–292
Ingolfsdottir K, Hylands PJ, Solberg Y (1986) Structure of vesuvianic acid from Stereocaulon species. Phytochemistry 25:550–553. https://doi.org/10.1016/S0031-9422(00)85527-2
Ingolfsdottir K, Gissurarson SR, Muller-Jakic B et al (1996) Inhibitory effects of the lichen metabolite lobaric acid on arachidonate metabolism in vitro. Phytomedicine 2:243–246. https://doi.org/10.1016/S0944-7113(96)80049-3
Ingólfsdóttir K, Gissurarson SR, Nenninger A et al (1997) Biologically active alkamide from the lichen Stereocaulon alpinum. Phytomedicine 4:331–334. https://doi.org/10.1016/S0944-7113(97)80042-6
Ismed F (2012) Phytochimie de lichens du genre Stereocaulon: étude particulière de S. halei Lamb et S. montagneanum Lamb, deux lichens recoltés en Indonésie. Universite de Rennes 1
Ismed F, Lohezic-Le Devehat F, Delalande O et al (2012) Lobarin from the Sumatran lichen, Stereocaulon halei. Fitoterapia. https://doi.org/10.1016/j.fitote.2012.09.025
Ismed F, Dévéhat FL-L, Rouaud I et al (2017) NMR reassignment of stictic acid isolated from a Sumatran lichen Stereocaulon montagneanum (Stereocaulaceae) with superoxide anion scavenging activities. Z Naturforsch C Bio Sci. https://doi.org/10.1515/znc-2016-0148
Johnson G (1938) The taxonomic importance and phylogenetic significance of the cephalodia of Stereocaulon. Ann Mo Bot Gard 25:729–769
Kirk P, Cannon P, David J, Stalpers J (2001) Ainsworth and Bisby’s dictionary of the fungi, 9th edn. CABI Publishing, Wallingford
Kokubun T, Shiu WKP, Gibbons S (2007) Inhibitory activities of lichen-derived compounds against methicillin- and multidrug-resistant Staphylococcus aureus. Planta Med 73:176–179. https://doi.org/10.1055/s-2006-957070
Lamb I (1951) On the morphology, phylogeny, and taxonomy of the lichen genus Stereocaulon. Can J Bot 29:522–584
Lamb M (1976) Structurally unusual types of cephalodia in the lichen Stereocaulon (subgen. Holostelidium). J Jpn Bot 51:1–7
Lamb M (1977) A conspectus of the lichen genus Stereocaulon (SCHREB.) HOFFM. J Hattori Bot Lab 43:191–355
Lamb M (1978) Keys to the species of the lichen genus Stereocaulon (SCHREB.) HOFFM. J Hattori Bot Lab 44:209–250
Lavergne R (1989) Plantes medicinales indigenes tisanerie et tisaneurs de la Reunion. Universite des Sciences et Techniques du Languedoc, France
Lee K, Yim JH, Lee HK, Pyo S (2016) Inhibition of VCAM-1 expression on mouse vascular smooth muscle cells by lobastin via downregulation of p38, ERK 1/2 and NF-κB signaling pathways. Arch Pharm Res 39:83–93. https://doi.org/10.1007/s12272-015-0687-3
Li B, Lin Z, Sun H (1991) The chemical constituents of four lichens from China. Yunnan Zhiwu Yanjiu 13:81–84
Magnusson A (1926) Studies on boreal Stereocaula. K Vet O Vitterh Samh Handl 30:1–89
Malik S, Pardeshi N, Seshadri T (1972) Chemical investigation of Indian lichens. Part XXX. Indian J Chem 10:1040
McCarthy P (2012) Checklist of the lichens of Australia and its island territories. In: Aust Biol Resour Study. http://www.anbg.gov.au/abrs/lichenlist/introduction.html. Accessed 10 Jan 2018
Millot M, Tomasi S, Sinbandhit S, Boustie J (2008) Phytochemical investigation of Tephromela atra: NMR studies of collatolic acid derivatives. Phytochem Lett 1:139–143. https://doi.org/10.1016/j.phytol.2008.07.004
Miyagawa H, Yamashita M, Ueno T, Hamada N (1997) Hypostrepsilalic acid from a cultured lichen mycobiont of Stereocaulon japonicum. Phytochemistry 46:1289–1291. https://doi.org/10.1016/S0031-9422(97)00501-3
Morita H, Tsuchiya T, Kishibe K et al (2009) Antimitotic activity of lobaric acid and a new benzofuran, sakisacaulon A from Stereocaulon sasakii. Bioorg Med Chem Lett 19:3679–3681. https://doi.org/10.1016/j.bmcl.2009.03.170
Myllys L, Hognabba F, Lohtander K et al (2005) Phylogenetic relationships of Stereocaulaceae based on simultaneous analysis of β-Tubulin, GAPDH and SSU rDNA sequences. Taxon 54:605–618. https://doi.org/10.2307/25065418
Natural Resources Conservation Service (2018) Classification for kingdom plantae down to genus Stereocaulon Hoffm. In: USDA. https://plants.usda.gov/java/ClassificationServlet?source=profile&symbol=STERE2&display=31. Accessed 23 Apr 2018
Nylander W (1860) Synopsis methodica lichenum. Martinet, Paris
Ogmundsdóttir HM, Zoëga GM, Gissurarson SR, Ingólfsdóttir K (1998) Anti-proliferative effects of lichen-derived inhibitors of 5-lipoxygenase on malignant cell-lines and mitogen-stimulated lymphocytes. J Pharm Pharmacol 50:107–115. https://doi.org/10.1111/j.2042-7158.1998.tb03312.x
Purvis O, Coppins B, Hawksworth D et al (1992) The lichen flora of Great Britain and Ireland. The British Lichen Society, London
Ramaut JL, Serusiaux E, Brouers M, Corvisier M (1978) Lichen acids of the Stereocaulon ramulosum group in Central East Africa. Bryologist 81:415–421. https://doi.org/10.2307/3242244
Rankovic B, Kosanic M, Stanojkovic T (2014) Stereocaulon paschale lichen as antioxidant, antimicrobial and anticancer agent. Farmacia 62:306–317
Riddle LW (1910) The North American species of Stereocaulon. Bot Gaz 50:285–304
Santiago K, Borricano J, Canal J et al (2010) Antibacterial activities of fructicose lichens collected from selected sites in Luzon Island, Philippines. Philipp Sci Lett 3:18–29
Seo C, Sohn JH, Ahn JS et al (2009) Protein tyrosine phosphatase 1B inhibitory effects of depsidone and pseudodepsidone metabolites from the Antarctic lichen Stereocaulon alpinum. Bioorg Med Chem Lett 19:2801–2803. https://doi.org/10.1016/j.bmcl.2009.03.108
Seshadri T (1944) A theory of biogenesis of lichen depsides and depsidones. In: Proceedings of the Indian Academy of Sciences—section A. Springer, India, pp 1–14
Sharma G (1997) Ethnomedicinal flora: ayurvedic system of medicine in a remote part of the Indo-Tibetan Himalayas. J Tenn Acad Sci 72:53–54
Singh R, Ranjan S, Nayaka S et al (2013) Functional characteristics of a fruticose type of lichen, Stereocaulon foliolosum Nyl. in response to light and water stress. Acta Physiol Plant 35:1605–1615. https://doi.org/10.1007/s11738-012-1203-8
Solberg Y (1977) Chemical investigation of the lichen species Alectoria ochroleuca, Stereocaulon vesuvianum var. pulvinatum and Icmadophila ericetorum. Z Naturforsch C Bio Sci 32:182
Solberg Y (1987) Chemical constituents of the lichens Cetraria delisei, Lobaria pulmonaria, Stereocaulon tomentosum and Usnea hirta. J Hattori Bot Lab 63:357–366
Stocker-Wörgötter E (2008) Metabolic diversity of lichen-forming ascomycetous fungi: culturing, polyketide and shikimate metabolite production, and PKS genes. Nat Prod Rep 25:188–200. https://doi.org/10.1039/B606983P
Vila J, Mollinedo P, Flores Y, Sterner O (2008) 1,3,7-trimethylguanine from the lichen Stereocaulon ramulosum. Rev Bol Quim 25:1–3
Vu TH (2014) Etude des acides gras du genre Stereocaulon etude phytochimique du lichen S. evolutum Graewe. Université de Rennes1
Vu TH, Le Lamer AC, Lalli C et al (2015) Depsides: lichen metabolites active against hepatitis C virus. PLoS ONE 10:1–14. https://doi.org/10.1371/journal.pone.0120405
Yamazaki M, Matsuo M, Shibata S (1965) Biosynthesis of lichen depsides, lecanoric acid and atranorin. Chem Pharm Bull (Tokyo) 13:1015–1017. https://doi.org/10.1248/cpb.13.1015
Yokota I, Shibata S (1978) A polysaccharide of the lichen, Stereocaulon japonicum. Chem Pharm Bull (Tokyo) 26:2668–2670. https://doi.org/10.1248/cpb.26.2668
Yokota I, Shibata S, Saitô H (1979) A 13 C-NMR analysis of linkages in lichen polysaccharides: an approach to chemical taxonomy of lichens. Carbohydr Res Res 69:252–258. https://doi.org/10.1016/S0008-6215(00)85771-7
Acknowledgements
The collaboration has been initiated in the frame of a Bioasia program (LICHENASIA 2009–2010) and further supported by the French Embassy in Indonesia. The authors also acknowledge A. Burel, V. Gouesbert and M.T. Lavault (Mric TEM. Univ. Rennes, BIOSIT—UMS 3480) for technical contribution in preparing lichens sections for light microscopy. J. Le Lannic is acknowledged for SEM images performed at CMEBA (ScanMAT, UMS 2001 CNRS—University of Rennes 1). A great acknowledgement is due to A. Chambet who provided some unvaluable documents and specimens from the Des Abbayes lichen Herbarium Collection and to JM Sussey for providing some sample of S. grande. Dr H Sipman (Berlin Museum) was also of great support with expertise in tropical lichen identification.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ismed, F., Lohézic-Le Dévéhat, F., Guiller, A. et al. Phytochemical review of the lichen genus Stereocaulon (Fam. Stereocaulaceae) and related pharmacological activities highlighted by a focus on nine species. Phytochem Rev 17, 1165–1178 (2018). https://doi.org/10.1007/s11101-018-9576-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11101-018-9576-y